
Key to Oak Diseases
"'What is an "identification key?' It is a road map of sorts that aids in the identification of a disease or an insect specimen observed in the field. It is based on a series of questions that have as answers choices between two or more alternative pathways. The choices describe symptoms or morphological characteristics thought to be most distinctive of a particular insect or disease. In order to use these keys, you must first decide or make an educated guess as to whether the damage or symptom is caused by an INSECT or DISEASE."
—from "Florida Tomato Scouting Guide", University of Florida.
The following key will help you identify common diseases of oak trees.
| 1. Foliage and/or twigs affected | go to #2 |
| 1. Trunk or branches affected | go to #8 |
| 1. Roots or root collar affected | go to #11 |
| 2. Leaf necrosis (dead spots or blights) present | go to #3 |
| 2. Necrosis absent; leaves affected but remain green | go to #6 |
| 3. Tiny to sprawling brown or gray spots along leaf margins and/or veins; no leaf blistering or puckering associated with spots | go to #4 |
| 3. Tan, crusty spots associated with leaf puckering; seen in the fall | view oak leaf blister |
| 3. Marginal, tip or interveinal leaf scorching; affectes all leaves on a branch | go to #5 |
| 4. Spots seen in the spring; leaves are tattered or distorted; twigs killed | view anthracnose |
| 4. Sprawling spots seen in the fall; often with yellow halo | view Tubakia leaf spot |
| 5. Scorching from leaf tip toward petiole; yellow to red line often precedes advancing scorch symptom on leaf | view bacterial leaf scorch |
| 5. Yellow, brown or dull green scorching from leaf tip toward petiole or beginning at leaf petiole; affected branch often wilts | view oak wilt |
| 6. Leaves and/or twigs with mycelium on upper or lower surface | go to #7 |
| 6. Light green, puckered areas in the spring; areas become necrotic in fall | view oak leaf blister |
| 6. Small, yellow pin-point spots on upper leaf surface; brown, hair-like projections on leaf underside | view fusiform rust |
| 7. Upper or lower surfaces of leaf with white, powdery growth; pale green spots may also be present when growth is absent | view powdery mildew |
| 7. Black, sooty or powdery growth typically on upper leaf surface | view sooty mold |
| 8. Branches or trunk galled or swollen | view crown gall |
| 8. Branches show dieback; cankers on trunk; tree in decline | go to #9 |
| 8. Branches show flagging, wilting; foliage yellows or browns | go to #10 |
| 9. Black, crusty, small spots or vertical stripes on trunk or branches | view Hypoxylon canker |
| 9. Conks or mushrooms protrude from trunk | view heart or canker rot |
| 9. Sour- or yeasty-smelling sap oozes from trunk; trunk stains dark | view bacterial wetwood |
| 10. Vascular discoloration present | view oak wilt |
| 10. Vascular discoloration not present | view bacterial leaf scorch |
| 11. Established tree in slow decline, white or thread-like material under bark | view Armillaria root rot |
| 11. Conks or mushrooms growing at root collar | view heart rot or wood decay |
| 11. Tree is newly transplanted; leaves turn brown, bark on lower stem may peel away | view root rot |
Oak Leaf Blister
Taphrina caerulescens
Symptoms
Oak leaves begin to show chlorotic, blister-like areas on the upper surface that can be as large as one half inch in diameter (Figures 1 & 2). The lower surface has gray depressions that correspond to the raised blisters. As the disease progresses, the blisters turn brown and the leaf will curl as the blisters coalesce. Premature leaf drop also may occur. Trees are not severely damaged, but the appearance of the tree may be unsightly. All oak species are susceptible to this disease.
 Figure 1. Chlorotic blister-like lesions.
Figure 1. Chlorotic blister-like lesions.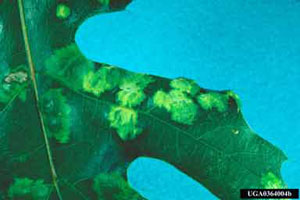 Figure 2. Puckered lesions of oak leaf blister.
Figure 2. Puckered lesions of oak leaf blister.Favorable Conditions
Leaves become infected when buds are beginning to open in the spring. The pathogen can survive winter on plant twigs and bud scales. The disease develops during wet, humid, mild conditions in the spring. Spores causing oak leaf blister are spread by wind and rain.
Control
Rake fallen leaves and debris and discard to reduce disease inoculum. No chemical control is necessary.
Anthracnose
Apiognomonia quercinia
Symptoms
Symptoms vary with host, weather and time of infection. Shoot blight is one of the first symptoms seen in spring. Blighting causes leaves and shoots to brown and shrivel. Young leaves become cupped or distorted with necrotic lesions. Large lesions often follow leaf veins or are delimited by leaf veins (Figure 3). Old lesions are papery and gray to white in color (Figure 4). On the underside of an infected leaf, tiny brown fungal fruiting bodies may be visible on or near major veins. Premature leaf drop is common. Mature leaves are fairly resistant and the symptoms are simple necrotic spots. If infection is severe, branch cankers and twig dieback can occur during winter and early spring. The symptoms usually first appear on lower branches and then spread upward.
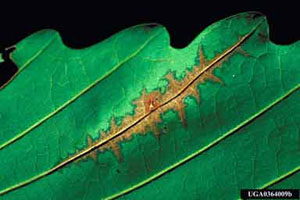 Figure 3. Anthracnose lesions following a leaf vein.
Figure 3. Anthracnose lesions following a leaf vein. Figure 4. Distorted leaves with papery lesions.
Figure 4. Distorted leaves with papery lesions.Favorable Conditions
Anthracnose fungi overwinter in twigs and plant debris. If winters are mild, the pathogen is active causing cankers and dieback. Spores are spread by wind and rain during the spring and infect new shoots. This disease can have multiple cycles per year if the weather is moderately warm and wet.
Control
Rake and destroy fallen leaves. Prune lower branches to increase air circulation and ensure proper tree fertility and irrigation. Chemical controls are rarely recommended unless trees are newly established.
Tubakia Leaf Spot (Actinopelte Leaf Spot)
Tubakia dryina
Symptoms
The symptoms caused by Tubakia are brown or reddish brown blotches on the leaves. Premature leaf drop and twig cankers are common if the trees are severely infected. Spots are well-defined on young leaves and enlarge to necrotic blotches on older leaves (Figures 5 & 6). Small fungal fruiting bodies can be seen within the lesions. Lesions may cause leaves to collapse if they are on veins and restrict water movement.
 Figure 5. Newly developed circular lesions caused by Tubakia.
Figure 5. Newly developed circular lesions caused by Tubakia. Figure 6. Brown lesions on older leaves of Tubakia leaf spot.
Figure 6. Brown lesions on older leaves of Tubakia leaf spot.Favorable Conditions
The disease overwinters on twigs and plant debris. It favors wet, humid conditions and warm temperatures. Primary spore dissemination is by wind and rain. Red oaks are more susceptible than white oaks.
Control
Rake and dispose of fallen leaves. Prune trees to increase air circulation and ensure proper fertility and irrigation.
Bacterial Leaf Scorch
Xylella fastidiosa
Symptoms
Symptoms of bacterial leaf scorch are described as marginal leaf burn and are very similar to drought stress symptoms (Figure 7). In addition to marginal leaf burn, there is a defined reddish or yellow border separating the necrosis from green tissue. Symptoms are more noticeable in late summer after hot, dry conditions, but symptoms can be expressed all year. Symptoms first appear on one branch or section of branches and on the oldest leaves. Each year symptoms will reoccur and progress to other parts of the tree (Figure 8).
 Figure 7. Marginal leaf burn due to bacterial leaf scorch.
Figure 7. Marginal leaf burn due to bacterial leaf scorch. Figure 8. Branch dieback due to bacterial leaf scorch.
Figure 8. Branch dieback due to bacterial leaf scorch.Favorable Conditions
The disease is spread by leafhoppers, spittlebugs and through root contact with neighboring trees. Since the pathogen is harbored within insects, warm temperatures and high populations of leafhoppers and spittlebugs are conducive for bacterial leaf scorch.
Control
Remove severely infected trees and replant using resistant species. Control weeds (to minimize insect populations) and ensure proper tree fertility and irrigation to maintain health and vigor.
Oak Wilt
Ceratocystis fagacearum
Symptoms
Oak wilt is rare in Georgia. The disease has not been positively identified at the UGA Plant Disease Clinic. Oak wilt is first observed near the top of the tree. Browning and bronzing of the leaves from the margins toward the petiole are the first symptoms of oak wilt. Eventually the leaves will drop prematurely and the tree will die. White oaks are moderately resistant to oak wilt. Red oaks often die within four weeks of the first symptoms. It may take years before other oaks die. The most reliable way to diagnose oak wilt on live oak is to observe leaf veins (Figure 9). Veins are chlorotic and eventually turn brown. Oak wilt on red oaks simply wilts young leaves and they turn pale green and brown. Mature leaves may have water soaking lesions or turn bronze on the leaf margins (Figure 10). Only individual branches or a portion of the canopy show symptoms. The fungus may cause brown streaking of sapwood (Figure 11). Red oaks may have fungal mats that break through the bark (Figure 12).
 Figure 9. Chlorotic veins on live oak.
Figure 9. Chlorotic veins on live oak. Figure 10. Bronzing of leaf margins on red oak.
Figure 10. Bronzing of leaf margins on red oak.
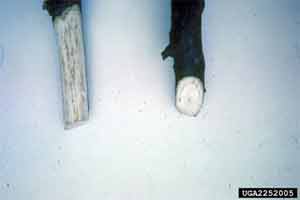 Figure 11. Brown streaking in sapwood.
Figure 11. Brown streaking in sapwood. Figure 12. Cracked bark due to fungal mats.
Figure 12. Cracked bark due to fungal mats.Favorable Conditions
The disease is spread by oak bark beetles and root grafts. It is most active during moderate temperatures. Red oaks are more susceptible than white oaks. The pathogen cannot tolerate temperatures above 90 degrees F ; so often, the pathogen dies in leaves and twigs. However, it may remain in the trunk and in the roots. Oaks are most susceptible in spring as new wood is forming.
Control
Remove infected trees. Trenching may be used to prevent root grafting. Trunk injections using a systemic fungicide have had some effect in reducing oak wilt spread within newly infected trees; however, this method is not recommended for trees in the southeastern United States because incidence is limited and of little value once disease is identified.
Fusiform Rust
Cronartium quercuum
Symptoms
Lower leaf surfaces often have inconspicuous yellow to orange pustules and black hair-like clusters (Figures 13 & 14). However, the orange pustules can become very noticeable in summer and fall as rust infection continues on oak leaves. Leaves may have yellow spots or necrotic blotches and develop premature leaf drop. Symptoms on oak are not severe, but it is the alternate host to a more devastating disease in pines.
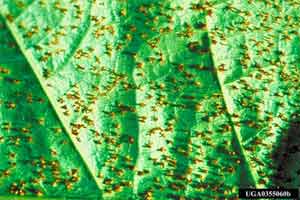 Figure 13. Yellow pustules.
Figure 13. Yellow pustules. Figure 14. Black hair-like clusters.
Figure 14. Black hair-like clusters.Favorable Conditions
Fusiform rust is most damaging in slash and loblolly pines. During the spring, pine galls produce spores that are wind-blown to young oak leaves. The pustules on the bottom of oak leaves then release spores in late spring that are carried by the wind to branch tips and tender, green pine needles. Most pine infections occur in April and early May. One spore stage (urediniospores) produced on oak leaves can continue to re-infect new oak leaves throughout the summer during warm, moist, humid conditions. This stage of the disease does not affect pines.
Control
Disease is usually insignificant to oaks, except in tree nurseries where the orange pustules can make leaves unsightly. Fungicides can reduce rust development on young newly planted oaks, but control is directed mostly towards saving pines.
Powdery Mildew
Erysiphe spp.
Symptoms
 Figure 15. White, powdery growth on the upper side of an oak leaf.
Figure 15. White, powdery growth on the upper side of an oak leaf.Powdery mildew is a white, powdery growth on the upper side of leaves (Figure 15). Tiny black fruiting bodies are usually present in late summer or fall. Infection may also occur in the buds and shoots causing a witches' broom as in the case of live oaks. Symptoms appear to be superficial.
Favorable Conditions
Powdery mildew survives the winter on fallen leaves and in the spring, wind and water disseminate spores to new leaves. Cool, moist weather conditions in the spring are ideal for infection. Powdery mildew prefers warm days around 80 degrees F and cool nights of 60 degrees F. Temperatures above 90 degrees F will reduce powdery mildew growth. Powdery mildew also requires high humidity but dry leaves. This pathogen usually attacks a crowded canopy with poor air circulation.
Control
Irrigate early in the morning, improve air circulation and remove fallen leaves. Witches' brooming may be pruned from the tree. Fungicides are generally not recommended or needed as the disease is not lethal to the tree.
Sooty Mold
Various fungi
Symptoms
 Figure 16. Black crust of sooty mold on foliage.
Figure 16. Black crust of sooty mold on foliage.Sooty mold is a crust-like or powdery black growth on the foliage and stems of infested plants (Figure 16). Sooty molds grow superficially and do not penetrate the leaves. Sooty mold fungi grow mainly on the excrement of sap-sucking insects such as aphids, scales, and whiteflies. Sooty molds do not attack the plant and it is mainly a cosmetic problem. Occasionally, severe build-ups can inhibit photosynthesis and reduce plant vigor. Sooty molds may persist long after the insects have left.
Favorable Conditions
Sooty molds are present where there is a high insect population. Spores are dispersed by water.
Control
Reduce insect population. No control is directed toward the sooty mold as its growth will stop when the insect population is controlled.
Crown Gall
Agrobacterium tumefaciens
Symptoms
Roots, stems and crowns form large, rough galls (Figure 17). Plants with large galls are often stunted and are the first to suffer from environmental stresses. Damage is greatest when galls encircle the root crown. Mature trees may survive, but severely stressed or diseased trees may be killed by secondary pathogens. Galls are initially spongy, becoming hard with age (Figure 18). Dead surface tissue will decay and slough-off.
 Figure 17. An extreme case of crown gall.
Figure 17. An extreme case of crown gall. Figure 18. Mature gall that is hard and rough.
Figure 18. Mature gall that is hard and rough.Favorable Conditions
A crown gall bacterium is dispersed in soil and water and enters through wounds on roots and crowns. Once inside, the bacterium transfers some of its genetic material into the trees cells. These cells form tumors and grow at a rapid rate. The bacteria may survive in the soil for two years and galls develop in temperatures near 72 degrees F. Temperatures above 86 degrees F stops normal cells from transforming into tumor cells but does not prevent galls after transformation.
Control
Avoid introducing crown gall bacterium into an area. Use resistant varieties and avoid injuring tree roots and trunks. Control insects that feed on stems and roots. Disinfect tools when pruning infected trees.
Hypoxylon Canker
Hypoxylon spp.
Symptoms
Yellowing and wilting of leaves often related to physiological stress may be the first symptom of this disease. Fungal mats (stroma) will develop beneath the bark of infected trees. Bark will begin to slough-off due to pressure from the stroma beneath it. Stroma exposed by sloughing bark is hard, tan to silver gray on the outside, and black within (Figures 19 & 20). There may be small patches to elongated strips. Old stroma eventually loses the gray color and appears black. The sapwood becomes tan to light brown and has a definite black border.
 Figure 19. Gray and black patches.
Figure 19. Gray and black patches. Figure 20. Silver gray patches.
Figure 20. Silver gray patches.Favorable Conditions
Hypoxylon cankers are opportunists that attack trees weaken by other factors such as heat, drought, wound, root injury or other diseases. Oak species most commonly infected are black, blackjack, laurel, live, post, and white oaks. Hypoxylon can be present as a latent colonist in healthy trees and this may account for the rapid invasion of stressed trees. The fungus is favored by warm temperatures of 60 to 100 degrees F but the optimal temperature is near 86 degrees F. Spores are primarily wind dispersed.
Control
Keep trees vigorous with proper fertility and irrigation. Prevent wounds from mechanical, weather and insect injury. Remove infected limbs or trees once they become hazardous to property or people, and to reduce disease spread to adjacent trees.
Heart or Canker Rot
Inonotus spp.
Symptoms
 Figure 21. Smooth, shelf-like conk.
Figure 21. Smooth, shelf-like conk.The first symptom of this disease is a white rot of the heartwood and the eventual death of the cambium and sapwood. The decay removes lignin and the wood becomes spongy. The basidiocarps or conks of Ionotus andersonni exhibits dull brown peg like structures that may exceed 20 inches in length on the outside of the tree. Conks of I. hispidus form in the summer and early autumn. The conks are smooth and shelf-like and are fully grown within one to two weeks (Figure 21). The top of the fungus is yellow to rusty red and the porous bottom is rust color. After three weeks, they dry and fall to the ground. Conks of I. dryadeus form at the base of infected trees among root flares. Conks are initially white or light-colored and turn black and crusty with age. Infected trees show symptoms of general tree decline including branch dieback, loss of leaves and yellowing or browning of leaves in summer. Trees weakened by drought stress, wounding or other injuries are most susceptible.
Favorable Conditions
 Figure 22. Cracked bark due to fungal mats.
Figure 22. Cracked bark due to fungal mats.Oaks in the red and black group are most often infected. Branch stubs within 16 feet of the ground are the most common infection sites, but entry also occurs through other injuries (Figure 22). Optimal temperature for growth is 95 degrees F. Old conks or remnants may continue to fruit for up to five years.
Control
Avoid unnecessary injury or stress to trees. Remove hazardous trees to protect property and people. Time pruning of infected branches to minimize exposure of susceptible tissues and when spores are not disseminated (late winter or spring).
Bacterial Wetwood
Symptoms
A sour odor is often associated with wetwood as water-soaked wood with large numbers of dead bacteria begin to break down. The build-up of bacterial populations within the tree causes fermentation resulting in internal gas pressure of up to 60 pounds per square inch. Foliage sometimes wilts and branches may dieback. However, most of the time, wetwood is a minor problem that leaves a vertical streak on the bark where pressurized liquid escaped out of wounds (Figures 23 & 24). Many times, secondary fungi and bacteria infect the surface liquid and create a slimy texture on the bark.
 Figure 23. Staining of bark.
Figure 23. Staining of bark. Figure 24. Bark stain due to wetwood.
Figure 24. Bark stain due to wetwood.Favorable Conditions
Bacteria that cause wetwood tolerate low oxygen and are often found in soils and on plant surfaces. Bacteria enter through assorted wounds above and below the soil line. The bacteria may lay dormant during the greatest periods of growth and become active in mature or older tissues.
Control
There are no known controls for bacterial wetwood. A 10 percent bleach solution may be used cosmetically to clean stains off the bark.
Armillaria Root Rot
Armillaria spp.
Symptoms
There are several general symptoms that accompany Armillaria root disease, including crown dieback, growth reduction, premature leaf drop or death of the tree. Because these fungi commonly inhabit roots, their detection is difficult unless characteristic mushrooms are produced at the base of the tree. Removing the bark will expose the characteristic, white mycelial rhizomorphs that grow between the wood and the bark (Figure 25). Short-lived mushrooms may be found growing in clusters around the bases of infected trees. They are honey brown to reddish in color (Figure 26). The fungus breaks down lignin and cellulose causing the wood to become spongy.
 Figure 25. White rhizomorphs of Armillaria.
Figure 25. White rhizomorphs of Armillaria. Figure 26. Honey-colored Armillaria mushrooms.
Figure 26. Honey-colored Armillaria mushrooms.Favorable Conditions
Spread occurs when rhizomorphs contact uninfected roots. Rhizomorphs can grow for distances of up to 10 feet and penetrate the roots by a combination of mechanical pressure and enzymatic action. Mushrooms are produced in late summer or autumn, and are most abundant during moist periods.
Vigorously growing trees often confine the fungi to localized lesions and limit their spread up the roots by secreting resin and rapidly forming callus tissues. But when infected trees are in a weakened condition, Armillaria spreads rapidly through the roots.
Control
Because these fungi are indigenous to many areas and live on a wide variety of plants and woody material, their eradication is not feasible. Management should be directed toward increasing tree vigor through proper irrigation and fertility.
Root Rot
Phytophthora spp.
Symptoms
Above-ground symptoms vary, but generally include reduced tree vigor and growth, yellowing or chlorosis of leaves and eventual collapse or death of the tree. Infected trees may decline slowly over one or more years, or they may collapse and die rapidly after resuming growth in the spring. Rapid death of trees usually occurs following excessively wet periods. On trees that decline slowly, leaves will yellow or brown while leaves on healthy trees remain green. To observe below ground symptoms, you need to remove several inches of soil around the base of the declining tree. A diagnostic reddish-brown discoloration of the inner bark and wood can be observed after cutting away the outer bark layer (Figure 27). Similar symptoms can be found on roots, but it is generally difficult to see root symptoms without removing the tree (Figure 28).
 Figure 27. Reddish brown wood discoloration.
Figure 27. Reddish brown wood discoloration.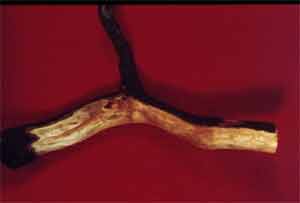 Figure 28. Brown discoloration of roots.
Figure 28. Brown discoloration of roots.Favorable Conditions
Phytophthora root and crown rots are caused by several Phytophthora species. While some species are much more destructive than others, all species require extremely wet or saturated soils in order to infect and cause significant damage. These fungi over-winter and persist in soil as mycelium in infected wood and can remain viable in the soil for years. When soils are wet, the fungus germinates and spreads to susceptible plant tissue where they infect. They may also spread to the soil surface and move over longer distances in runoff water. Trees appear to be most susceptible during spring and autumn, which are also the times of year when soil temperatures are most conducive to fungus growth and zoospore production. Fungal activity is low in the winter when trees are dormant.
Control
Control of Phytophthora root rot is most successful using an integrated program of cultural practices including good soil drainage and proper irrigation. Avoid planting trees too deep as this also contributes to disease development and decline.
References
Forestry Images: www.forestryimages.org
Purdue University: ag.purdue.edu/department/btny/ppdl/
Texas A&M University: plantclinic.tamu.edu
University of Minnesota: pdc.umn.edu
University of Natural Resources and Applied Life Sciences: boku.ac.at/en/
West Virginia University: www.davis.wvu.edu
Status and Revision History
Published on Aug 15, 2005
Published on Feb 10, 2009
Published on May 05, 2009
Published with Full Review on Jun 22, 2012


























































