- How Do Adjuvants Work?
- Today's Adjuvant Menu
- Adjuvants versus Surfactants
- Other Adjuvants
- New Product Classes
- So Why Not Just Use Common Kitchen Soaps and Detergents?
- Natural Surfactants
- Bottom Line?
- Additional References
- Adjuvant Retailers
Overcoming pesticide water solubility issues was a monumental task in the 1950s and 60s. Most pesticides were not formulated to use water as their carrier. Today, however, the majority of pesticides are formulated to use water. The waxy surfaces of many insects, fungi, and plants make it difficult for most water-based spray solutions to penetrate their target. To overcome this barrier, adjuvants have been developed. Adjuvants can determine how well a pesticide works but, when used inappropriately, they can cause crop damage. Adjuvant is a broad term describing any additive to a spray tank that enhances pesticide activity. Examples of adjuvants are surfactants, spreader stickers, crop oils, anti-foaming materials, buffering agents, and compatibility agents. Surfactants are adjuvants that facilitate and accentuate the emulsifying, dispersing, spreading, wetting, or other surface modifying properties of liquids.
Many pesticides require the addition of an adjuvant, and some do not. When applying fungicides, insecticides or herbicides without a recommended adjuvant, 30 percent to 50 percent reduction in pest control can be expected. Adjuvants may cause damage to a plant if the wrong adjuvant is used or if it is used at too high a concentration. Using the correct adjuvant on a greenhouse crop is a critical decision. This bulletin is intended to describe how adjuvants differ and what adjuvants are best to use.
How Do Adjuvants Work?
To understand how adjuvants work, it helps to understand how water works. Each water molecule is bipolar, meaning it has a negative and a positive charge, very much like a magnet. If you put several water molecules together, the positive and negative forces attract each other (Figure 1) .
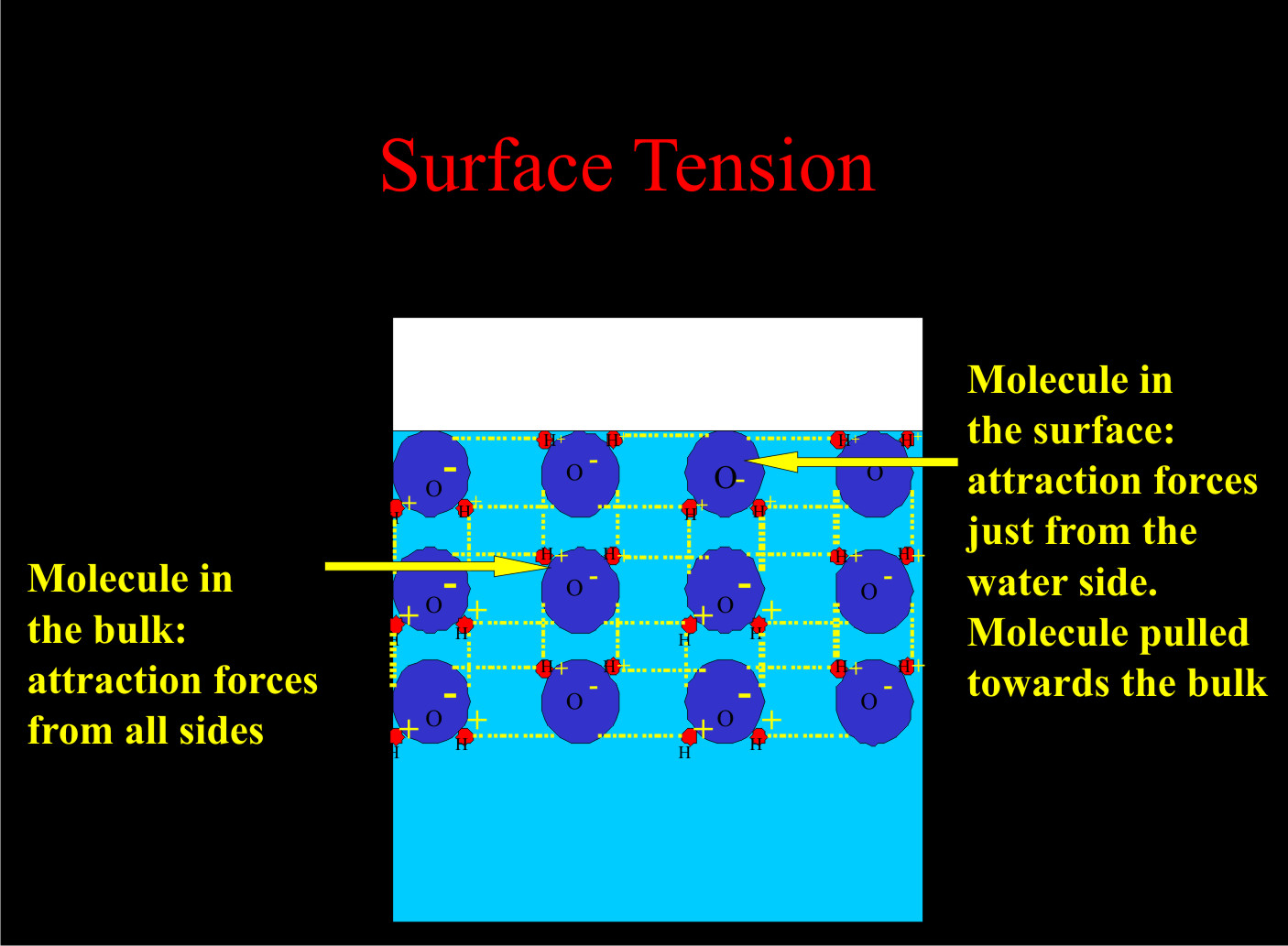 Figure 1. The interacting charges of water molecules (oxygen represented in blue - negatively charged and hydrogen represented in red - positively charged).
Figure 1. The interacting charges of water molecules (oxygen represented in blue - negatively charged and hydrogen represented in red - positively charged).
The molecules on the surface of a water droplet are held together with more force than those of the interior water molecules. This causes surface tension, which can prevent many things from going into solution and getting wet. Surfactants overcome surface tension. Most surfactants have a water-loving polar head (hydrophilic head) and water-hating non-polar tail (hydrophobic tail) (Figure 2).
These components of a surfactant molecule help break water surface tension, allowing the pesticide to be more evenly dispersed on a surface and to reach its target. When water molecules come into contact with unlike substances, several things may happen. If the substances have a similar charge, the two forces repel each others. If they have different charges, the two forces will attract each other. If there are no charges, there will be no reaction. Water, when placed on most hydrophobic surfaces, will bead. This beading is caused by surface tension, and this surface tension can be reduced by the addition of surfactants (Figure 3).
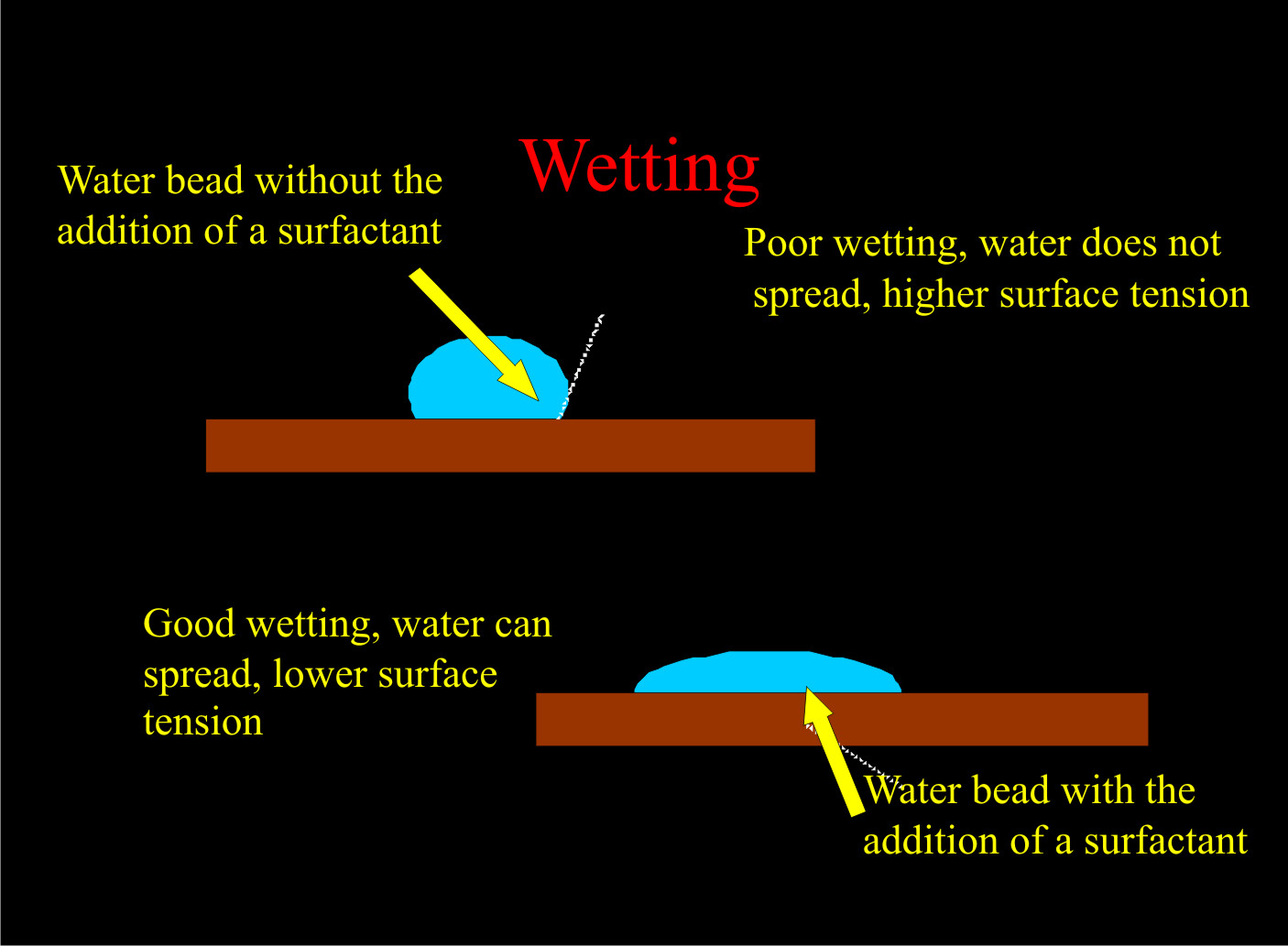 Figure 3. Water beads and how they are affected by surfactants.
Figure 3. Water beads and how they are affected by surfactants. Figure 4. Horticultural oil / dishwashing liquid damage to pansies.
Figure 4. Horticultural oil / dishwashing liquid damage to pansies.
The lower the surface tension in a pesticide solution, the better the pesticide coverage, allowing more pesticide to reach its target.
It’s important to know which adjuvant is best. For instance, one day we received a panicked call from a grower who indicated that his pansy crop was in trouble. Earlier that day he discovered his crop had a serious aphid infestation. He sprayed thoroughly, but he soon found that his efforts were futile, as the spray droplets ran off the foliage. Rather than investigate or consult, the grower decided to use horticultural oil with the insecticide and spray again. It was late in the afternoon, hot and sunny. By 5 p.m.. the leaves of the pansies started wilting. Panicked, he decided to apply a commonly used household dishwashing liquid solution to the pansies to rinse off the oil, knowing soaps dissolve oils.
By the time he called for assistance, 1,300 flats of pansies were already dissolving into small blobs of dark, black-green slime (Figure 4).
 Figure 5. Horticultural oil damage to pansy flower and foliage.
Figure 5. Horticultural oil damage to pansy flower and foliage.Had the grower read the label, the horticultural oil he used clearly indicated it was a penetrating type oil to be used only on fruit crops, ornamental trees and shrubs when they are dormant (not on actively growing foliage). In hot weather oils can be very damaging to plants (Figure 5).
This unfortunate incident could have been prevented if the person applying the pesticide had taken time to read the label. When explosive thrips and viral epidemics occur or insect resistance to pesticides increases, many growers are desperate and easily tempted to use any handy surfactant wetting agent. The science of adjuvant technology has increased the effectiveness of our pesticides, but only when used properly. It is critical that a pesticide be teamed with the right adjuvant.
Today’s Adjuvant Menu
The following is a general review of the types of adjuvants on the market, including how they work and what damage may occur when they are not used properly. For specific information on greenhouse-labeled products, please contact your county extension agent or manufacturer and read the labels carefully before using them. Today, there are close to 400 adjuvant related products including surfactants, wetting agents, and other adjuvant related products on the market, and only a small percentage of these are suitable or labeled for greenhouse use.
Adjuvants versus Surfactants
Surfactants
Surfactants are adjuvants that reduce surface tension within the external surface layers of water. There are four different types of surfactants:
Anionic Surfactants are negatively charged, and enhance foaming and other spreading properties. For example, shampoo for hair contains sodium or ammonium laureth sulfate, which is the preferred anionic surfactant for hair. Using an anionic surfactant in the greenhouse can cause problems with sprayers that have an agitator, or any system where the foam could disrupt water flow or pump suction.
Cationic Surfactants are positively charged, and are often very toxic to plants as they can disrupt membrane ion balance. Cationic surfactants are not widely used for pest control, but they are more commonly used in heavy-duty cleaning compounds. Don’t grab a bottle of engine wash surfactant used to clean the tractor and add it to your pesticides when spraying Easter lilies. The results can be devastating.
Amphoteric Surfactants are unusual in that they will form either a positive or negative charge in water, depending upon the pH of the solution. Their use in horticulture crop protection is rare. These products are used very specifically to match the properties of specific pesticide formulations to carrier components or other materials, and are generally not available for use in the greenhouse as a stand-alone product.
Nonionic Surfactants do not have a charge in solution and are the most commonly used surfactants for the horticulture industry. When used properly, they as a class do not harm plants, remain stable, and do a good job of breaking water surface tension. However, application rate is critical. When applied at too high a rate plant injury may result (Figure 6).
 Figure 6. Nonionic surfactant damage to pansy (Viola spp.), Note regrowth.
Figure 6. Nonionic surfactant damage to pansy (Viola spp.), Note regrowth. Figure 7. The effect of excess soil wetting agent on pansy (Viola spp.) foliage.
Figure 7. The effect of excess soil wetting agent on pansy (Viola spp.) foliage.
Nonionic surfactants are now being researched in relation to controlling fungal infection, especially in hydroponic systems. When a fungal zoospore is coated with a nonionic surfactant, it does not function well. Dr. Ann Chase (pathologist at the University of Florida) and others are currently reviewing the effect of these products on fungi.
Wetting Agents are commonly mixed in growing media. They allow the media, such as peat moss, which is often hydrophobic (water-hating) when dry, to accept water into its collective structure. Wetting agents work much like surfactants, breaking the surface tension of water and helping the water transfer from particle to particle. However, their chemistry is different from most surfactants. Many are made up of polyoxyethylene esters, ethoxy sulfates, or derivations of these components mixed with nonionic surfactants.
When applied at too high a rate, a wetting agent can cause membrane permeability problems and cause toxicities that kill roots and leaves (Figure 7).
Occasionally, media from commercial companies contain too much wetting agent. This can cause disfigured, off-color, slow growing plants. Temperature and weather can affect how the plant will react to the presence of a large amount of wetting agent in bark or peat soils.
Other Adjuvants
Penetrants dissolve or penetrate waxy layers on leaves and allow other chemicals to interface with plant cells or insect epidermal tissue and enter the spaces between the epidermal cells, sometimes called “cell free space.” Penetrants can contain petroleum by-products, crop oils, complex alcohols, and other hydrocarbon-based materials. They work well under very specific conditions and with very specific crops grown outdoors. In the greenhouse, stand-alone products are rare.
Anti-condensates
Greenhouse walls made of various films, especially in poly houses with poor ventilation, have condensate problems and drip in the most inopportune places. Anti-condensates act like surfactants, making it easier for water to spread throughout the surface of the film instead of collecting as droplets on the plastic and dripping on plants. By creating a film, the water can be carried off as a thin sheet along the pipe or plastic film. They work great, but should never, ever be used on plants or on soil.
Thickeners are used to reduce drift of sprays
They may contain polyacrylamide, polyethylene polymers, polysaccharides (long-chain sugars), or vegetable oils. These products not only help volatile pesticides become less volatile, they also cause the carrier solution to become more viscous and heavier. This is beneficial when spraying an outdoor production area near a housing subdivision. It reduces drift, odor, and waste. However, when thickeners are applied at too high a dosage, they can cause surface deposits on foliage, excessive exposure to the pesticide, or clog spray nozzles. Use these adjuvants only in outdoor situations and only after reading the label for information about compatibility and phytotoxicity.
Emulsifiers
Emulsifier agents work by coating tiny particles or groups of the liquid molecules and preventing them from coagulating with other like molecules. Unlike oil and vinegar, the emulsifiers allow oil and water solutions to mix. These products are usually added by the manufacturer and are commonly mixed with petroleum-based pesticides to help them mix more readily with water.
Spreader and Stickers
Spreaders are compounds that cause the surface tension of the pesticide to be reduced in such a way that it easily spreads into a very thin film over a surface. Like surfactants, spreaders and stickers increase the efficiency of the pesticide dramatically. They may contain fatty acids, latex, aliphatic alcohols, crop oils such as cottonseed, or inorganic oils. Each formulation is very different.
Stickers are very much like thickening agents or oils in that they cause the pesticide solution to adhere to the leaf surface, resisting rain, evaporation and runoff. Some products use emulsified polyetheylenes, others use polymerized resins, fatty acids or petroleum distillates. Stickers are commonly used in field crops (like corn and soybeans) where residue on leaves is not a problem. However, in greenhouses, they can cause a mess, especially on leaves with indentations such as Pileas, or hairy plants such as Dusty Miller. Some spreader/sticker combinations cause phytotoxicity in tender annuals and herbs.
Oils
There are two types of oil: crop oils that are derived from soybean and other crops, and inorganic oils that come from petroleum refineries. Some are used as suffocates, which cover the insect and cut of its air supply. Others are used as penetrants to break apart the chitin layers (such as with scales) and cause the insect to die. Still others are used like surfactants, forming film over the leaf, breaking down the cuticle, and allowing the pesticide to enter its target. Some oils are blended with surfactants, to take advantage of the properties of both adjuvants. Oils are rarely recommended for greenhouse use.
New Product Classes
Alkyl polyglucosides. As complex as this term sounds, it simply refers to modified sugars. It has been found that some modified sugar molecules have surfactant-like properties and may be used as spreader/stickers. Although this class of adjuvant has been around for some time, it is only now becoming popular due to its organic origins, biodegradability, and the fact that it is environmentally friendly. There are low foaming and high foaming types, and many are very good wetting agents. Originally developed as environmentally safe cleaning agents (car wash cleaners) in England, they have transitioned to being used in laundry detergents and as agronomic adjuvants. These products have very low potential for phytotoxicity because they are derived from plant sugars. Research on these materials is on-going. Current work indicates they work well with glyphosate herbicide.
 Figure 8. Organosilicone surfactant damage to pansy (Viola spp.) leaves when applied in hot (>90ºF) sunny weather.
Figure 8. Organosilicone surfactant damage to pansy (Viola spp.) leaves when applied in hot (>90ºF) sunny weather.Organosilicates are not exactly new; they were developed in the 1970s, and have many uses including silicone-based sprays for waterproofing. About 10 years ago, their use as spray adjuvants for crop production was discovered. Organosilicate surfactants are very good at increasing the “rainfastness” of pesticides. They also reduce the surface tension and allow everything from micronutrients to fungicides to enter the leaf stomates. These products have phenomenal wetting abilities. Depending on the pesticide they are used with and the application rate, results may be good or bad, so be careful when using these products. Phytotoxicity can occur if these products are used at too high a rate or used when temperatures are above 90 degrees F. Some research suggests that the wetting properties of these surfactants are so good that they can also allow bacteria and fungi to more easily invade plants (via stomata). A sanitary facility, careful applications, and rotation with other surfactants is required to realize the benefits with these products. Applications of organosilicone surfactants in 90+ degree F weather can cause plant damage (Figure 8).
Inorganic Salts
There is continuing research on the use of certain inorganic salts as surfactants. However, most of this work is related to herbicide applications. Many of these products are salts of sodium, potassium and ammonium. Others contain calcium, iron, zinc and magnesium salts and are used in industrial cleaning agents and adjuvants. They also cause plant problems, particularly on greenhouse crops.
So Why Not Just Use Common Kitchen Soaps and Detergents?
There are very few adjuvant “soaps” on the market today. Most soaps are in bars and are used for bathing. Also, soaps react with cations in soil and fertilizer and can leave a precipitate residue on foliage. Most greenhouse workers remember gray precipitate on foliage resulting from soap residue.
Today, dishwashing liquids contain both anionic and nonionic surfactants. In most circumstances, their combined effects are synergistic and very potent. None are labeled for use on plants.
Natural Surfactants
Natural surfactants are biodegradable, wetting agents and oils that are processed differently from “crop oils” and alkylated sugars. Materials such as coconut oils, palm oils, castor oils, lanolins, wheat amino acids, and many others have been used in the past, but there is little research to verify these products are effective when used in combination with pesticides or in a greenhouses environment. Furthermore, there is evidence that these products may serve as food sources for bacteria and fungi. Until research gives us confidence in these products, exercise caution when using them.
Bottom Line?
Certain pesticides are recommended to be applied with oils, others with only non-ionic surfactants. The pesticide label specifies whether an adjuvant is needed and which class of adjuvants to use. These recommendations are strict for a reason. Experimenting with surfactants is a risky business. If, after reading the label, you are still unclear about which adjuvant to use, ask the company that produces the pesticide. They will tell you if a surfactant is present in the pesticide or which surfactant products can safely be used with the pesticide. There has been much research conducted with adjuvants and adjuvant / pesticide interaction. Most pesticides have specific requirements. So after you spend a few hundred dollars for a pint of pesticide, ask yourself, “Do I want this product to work?” Add the recommended adjuvant!
Additional References
Baird, J.V., and J.P. Zublena. 1993. Soil Facts: Using wetting Agents (Nonionic Surfactants) on Soil. North Carolina Cooperative Extension Service Publication. AG-439-25.
Irish, B.M., J.C. Correll, and T.E. Morelock. 2000. The Effect of Surfactants On White Rust (Albugo occidentalis) of Spinach. Texas A & M University Horticulture, Web site Abstract. Pp 1.
Jordan, T. N. 2001. Adjuvant Use With Herbicides: Factors To Consider. Purdue University Cooperative Extension Service Publication WS-7. Pp 1-5.
McCarty, B. 2001. Wetting Agents. Clemson University Cooperative Extension Service Publication. Pp 1-2.
Stanghellini, M.E., and R.M. Miller. 1997. Biosurfactants: Their identity and Potential Efficacy in The Biological Control of Zoosporic Plant Pathogens. Plant. Disease. 81:4-12.
Thompson-Harvey, L. 1991 A Guide To Agricultural Spray Adjuvants. Fresno, California: Thompson Publications. Pp. 1-20.
Adjuvant Retailers
Below is a list of a few distributors and retailers of adjuvants at the time of this publication release. Consult the World Wide Web for additional adjuvant availability.
| Company | Phone | Web Address |
| A. M. Leonard | 1-800-543-8955 | www.amleo.com |
| Ben Meadows Company | 1-800-241-6401 | www.benmeadows.com |
| Do My Own Pest Control | 1-866-581-7378 | www.domyownpestcontrol.com |
| Forestry Suppliers, Inc. | 1-800-647-5368 | www.forestry-suppliers.com |
| Gempler's | 1-800-382-8473 | www.gemplers.com |
| Helena | 1-901-761-0050 | www.helenachemical.com |
| Hummert International | 1-800-325-3055 | www.hummert.com |
| Lesco | 1-800-321-5325 | www.lesco.com |
| Scott's Professional Products | 1-800-492-8255 | www.scottsprohort.com |
| Southern States | 1-804-281-1000 | www.southernstates.com |
| Waldo & Associates | 1-800-468-4011 | www.waldoinc.com |
Trade and brand names are used only for information. Cooperative Extension, University of Georgia College of Agricultural and Environmental Sciences does not guarantee nor warrant published standards on any product mentioned; neither does the use of a trade or brand name imply approval of any product to the exclusion of others which may also be suitable.
Status and Revision History
Published on Feb 05, 2007
In Review on Jan 05, 2010
Published on Jan 08, 2010
Published with Full Review on Jan 22, 2013
