- History, Uses and Botanical Classification and Development
- Carrot Cultural Management
- Variety Selection
- Soil and Nutrition Management
- Disease Management
- Insect Management
- Managing Weeds in Carrots
- Irrigation Management for Carrots
- Pesticide Application
- Fumigation Equipment
- Good Agricultural Practices in the Harvest, Handling and Packaging of Fresh Carrots
- Harvesting Carrots
- Marketing Carrots
- Production Costs
Foreword
This publication is the result of a joint effort among the seven disciplines in the University of Georgia College of Agriculture and Environmental Sciences that serve the Georgia carrot industry. It is the most comprehensive production guide for carrots in Georgia ever assembled. The 13 topics covered in this bulletin are all integral parts of a successful carrot management program. Each topic is designed to focus on a particular aspect of production and provide the latest management technology for that phase of production. It is hoped that the information contained in this publication will assist growers in improving profitability in carrot production. Chemical pest control recommendations are subject to change from year to year and thus, only general pest control guidelines are mentioned in this publication. Growers are urged to consult the current Georgia Pest Management Handbook or check with their local county extension agent regarding the most recent chemical recommendations. Mention of trade names in this publication is not an endorsement of a particular product nor a lack of endorsement for similar products not mentioned.
Acknowledgments
The authors would like to express their gratitude to the following persons without whose help this publication would not have been possible: Mr. Rick Reed - CEC, Coffee County, Mr. John Ed Smith - CEC, Pierce County, Mr. Bob Boland - CEC, Brantley County, Mr. Randy Franks - CEC, Wayne County, Mr. James Reid - CEC, Jeff Davis County, Mr. Danny Stanaland - CEC, Bacon County, Dr. Don Sumner - Dept. Plant Pathology, Dr. Ron Gititas - Dept. Plant Pathology, Dr. Estes Reynolds - Dept. Food Science and Technology, Mr. Anthony Bateman - Dept. Horticulture, Miss Pam Lewis - Dept. Horticulture, Mrs. Jan Howell - Dept. Ag Engineering, Mrs. Kay Dunn - Dept. Horticulture, Mrs. Soccoro Seela - Dept. Entomology, Mrs. Priscilla Dolney - Dept. Ag Engineering, Mrs. Alice Pitts - Dept. Agricultural and Applied Economics.
History, Uses and Botanical Classification and Development
Carrot production in Georgia is a relatively new industry. Commercial production was extremely sparse until the early 1990s with only a handful of acres, mostly grown for the processing market. The original areas of production centered around Macon and Colquitt Counties. Fresh market production in Georgia originated primarily in Pierce County. Production then spread to areas in Wayne, Brantley, Coffee and Jeff Davis Counties and is still active in most of these counties. Today interest in both fresh market and processing carrots extends through-out the lower two thirds of the state.
Carrot (Daucus carota var. sativus) is a member of the Umbelliferae family. Other vegetable crops and herbs in this family include celery, parsnip, parsley, dill, caraway, anise, coriander and fennel. Domestic carrots may have evolved from a wild form similar to its relative known in North America as Queen Anne's Lace. The family name comes from the flower form, which is an umbel. Characteristic of most of the family's plants, an umbel has individual flower stalks originating from the same point on the stem.
Carrots probably originated in Asia around northwest India. Cultivation of carrots for medicinal purposes began 2000 to 3000 years ago. They were used for a myriad of medicinal purposes including stomach ulcers, abscesses, bladder, liver and kidney problems, to aid in childbirth and even as aphrodisiacs. Cultivation of roots for consumption dates back to 600 A.D. when purple root types were grown in the area currently known as Afghanistan.
Yellow types were eventually selected and produced in Syria and Iran in the ninth or tenth century. Carrots were introduced to China by the thirteenth century and cultivation spread from the Middle East to Italy, Spain and throughout Europe by the fourteenth century. Eventually, white and orange types were selected. Orange types, first grown in the Netherlands during the seventeenth century, were brought to North America by early settlers. The root was popular with Native Americans and production currently exists worldwide.
One of the reasons production is so widespread is that carrots are the major single source of Vitamin A in the diets of many cultures. They are also a good source of other vitamins, minerals and fiber. Carrots are produced for a variety of uses. Fresh market production for retail sales is still an important market. Fresh packed articles include peeled baby carrots, carrot sticks, shredded carrots and salad mixes. Processing markets include baby food production, frozen and canned products. Carrots are popular as snack foods, for deli trays, in salads, cooked in casseroles, as main vegetable dishes as well as numerous other culinary creations.
Carrot varieties produced in the United States are biennial in nature, requiring two full seasons from germination to seed production. However, it is grown commercially as an annual for its large storage root produced during the first year. True annual forms of the crop also exist but are not produced in the United States. Young carrots are characterized by a tight rosette of finely serrated leaves and a slender tap root with fine fibrous side roots. As the tap root enlarges and reaches maturity, the conductive tissue is distinguished as a light-colored core (xylem) and the deeper orange cortex (phloem).
Carrot Cultural Management
Climatic Requirements
Small carrot seedlings up to six leaves cannot with-stand hard freezes but are somewhat frost tolerant. Optimum temperatures are in the range of 60-70 degrees F, with daytime highs of 75 degrees F and nighttime lows of 55 degrees F ideal. Although the crop can be grown outside this range with little or no effect on tops, temperatures differing drastically from the above can adversely affect root color, texture, flavor and shape. Lower temperatures from this range may induce slow growth and make roots longer, more slender and lighter in color. Carrots with a root less than one inch in diameter are more susceptible to cold injury than larger roots.
Hot, sunny days can injure or kill young plants. Long periods of hot weather may depress carrot yields, cause strong terpinoid flavor and bitter taste in roots, and result in atypically short and blunt roots. Disease pressure also increases as temperatures warm in late spring and summer. Carrots need an ample supply of moisture from rainfall and/or irrigation throughout the growing season since they are not drought tolerant. A consistent moisture supply helps keep the crop growing and reduces the incidence of splitting from growth flushes.
Although technically biennial in nature, carrots can produce seedstems prematurely in Georgia. This is commonly known as bolting. Most carrot production in Georgia occurs during the fall months, through winter and into spring. Cool winter months followed by increasing daylength in spring can result in carrots being exposed to conditions that induce flowering. Flower induction in carrots requires a period of six to eight weeks of temperatures below 50 degrees F. Increasing daylength accelerates flower induction. Once flowering is initiated, the seedstalk elongates rapidly, particularly as temperatures increase. Although fall heat at planting is not injurious if they are well watered, carrots will tend to produce a higher percentage of seed stems as spring warming turns into summer heat. Carrots that have produced seed stems are not marketable.
Planting and Spacing
Carrots are exclusively direct-seeded. Most carrots in Georgia are planted from August 15 through October 31. Carrots produced in north Georgia would require a spring planting date. Spring plantings in south Georgia have not proven to be as successful as fall plantings. However, spring dates from early January through early February in south Georgia and late February through March in north Georgia would be appropriate. Plantings in August will require frequent irrigation during periods of hot, dry weather. Ideal planting conditions are more prevalent from late August to late October. Plantings from November to mid December have been successful; however, the risk of freeze damage to later planted carrots increases as planting dates are delayed. Carrots planted in August through December will be harvested in December through May in most years.
Spatial arrangements for planting can differ, although basic spacing is the same in each scenario. Carrots can be successfully planted with vacuum, belt or plate seeders. Often a special attachment called a scatter plate or spreader shoe is added to the plate planters to scatter the seed in a narrow band. Ideal patterns are twin rows that are 2.5-3.5 inches apart. Three or four of these twin rows are situated on one bed, depending on the width of the bed. One arrangement is to plant three twin rows on beds that are on 72-inch centers. The other arrangement currently used is to plant four twin rows on a 92-inch bed (center to center). The sets of twin rows are 14 to 18 inches apart. Beds on 72-inch centers will have approximately 48 inches of formed bed. Row spacing wider than 18 inches will reduce total plant stand per acre and thus, will reduce total yield. Ideal plant populations should be in the range of 450,000 for fresh market carrots and 300,000 for processing carrots. Beds that are slightly raised are advantageous because they allow for good drainage.
Other arrangements for planting may be adapted and work equally as well. High density arrangements with rows in sets of three (instead of two) are also used. In this arrangement, carrots are seeded in six sets of triplet rows with approximately two to three inches between rows in the triplet set. There is also minimal space between the sets -- around four inches. This arrangement is planted without raised beds, and the rows are almost continuous with no distinguishable break except the width of the tractor tire. The population density of this arrangement will be considerably higher than the other arrangements described. Disadvantages of high density plantings can include producing fewer jumbo carrots and lack of airflow through the field that can increase the incidence of foliar diseases. Also, planting without slightly raised beds is only recommended on very light and extremely well-drained soils.
Carrots should be spaced 1.5 to 2 inches apart within the row. Carrot seed should be planted no deeper than 1/4-1/2 inch. A final stand of 14 to 18 plants per foot of twin row is ideal. Beds should be firmed and not freshly tilled before planting, and soil should be firmed over the seed at planting. A basket or roller attachment is often used to firm the soil over the seed as they are planted. Seeds should be planted one-quarter to one-half inch deep. Light irrigation will be required frequently during warm, dry periods for adequate germination.
Windbreaks are almost essential in areas with primarily sandy soils. Sand particles moved by wind can sever young carrot plants, severely reducing stand. Small grain strips planted between beds or at least planted between every few beds can help reduce this sandblasting injury.
Variety Selection
There are four basic types of carrot cultivars that are generally distinguished by the shape of the root. Within each type there are many different varieties to select from. The carrot root consists of the phloem (or cortex) and the xylem (or core). More desirable varieties have a thicker phloem and a minimum of xylem. The phloem is deeper orange and flavorful. The xylem tends to be paler, tasteless and woody.
Taper-Rooted or Pointed
These roots decrease gradually in diameter from crown to tip and taper to a point. The Danvers and Imperator types of carrots fall into this category.
Danvers varieties are classified as half long and taper slightly in a conical shape from 2 to 2.5 inches in diameter at the crown to a length of 6 to 7 inches at maturity. Danvers carrots are deep orange with a light center. They have excellent quality early but can become fibrous with age.
Imperator varieties are probably the most widely grown fresh market type. They have a long tapered tip from a shoulder diameter of 1.5 to 2 inches to a length of 7 to 12 inches. Imperator types have deep orange phloem and light orange xylem and can become woody at full maturity. Young roots have excellent quality, however.
Stump-Rooted or Blunt
As in the taper rooted types this root type also tapers from crown to tip but the tip is blunt instead of pointed. The Chantenay types belong to this group.
Chantenay carrots are 2 to 2.5 inches in diameter at the crown and have a short, conical shape. These roots are shorter than Imperator or Danvers types, growing to a length of 4.5 to 5.5 inches with a medium to large neck. Chantenay types have medium to light orange phloem with a light to red core. Since they have coarse texture in raw form and produce a relatively small amount of waste they are popular as processing carrots and are the most widely grown.
Cylindrical
This type vary little from crown to tip and are somewhat blunt at the tip. Nantes type carrots are defined by this group. Most home garden cultivars are of this type. As per the name of the type these carrots are virtually cylindrical in shape. They have a blunt end with a shoulder diameter of 1.5 inches and a length of 6 to 7 inches. The thick phloem is bright orange and the xylem is often hardly noticeable. They have little top growth but excellent quality.
Ball-Shaped
Among this type of carrots are both round and stump-rooted varieties. The Amsterdam types have roots 0.5 to 0.75 inches in diameter and 2.5 to 3 inches long. Many are used for baby carrots, mixed vegetables and freezing. The more round shaped varieties, which are about 1.5 inches in diameter and length, perform well in heavy soils. They are best when harvested before full maturity and must be hilled to prevent green shoulders.
Variety Selection Criteria
Commercially grown carrot varieties in Georgia are generally of the Imperator type. Numerous commercial varieties exist, however, there are vast differences in performance of varieties over varying environmental conditions. Qualities that are important for commercial carrot production include yield, color, top growth, core diameter, length and uniformity. Top growth and low bolting are critical for carrots grown during fall and winter in Georgia.
Although yield is an important criteria, it should not be the only selection criteria. Good top growth is essential since most mechanical harvesters utilize the tops to extract carrots from the soil. Rich orange color and a small diameter core are essential fresh market characteristics. Acceptable length, uniformity and maturity are also important for packing efficiency and maximum yield. Certainly good flavor and high sugar content are desirable characteristics and are a marketing point with Georgia carrots. However, flavor is not always the most important factor in variety selection as visual appearance, yield and harvesting and packing factors often take priority. Basically a variety must be adaptable to the area, produce a competitive yield and be acceptable to buyers. Although disease resistance is not a major factor in carrot varieties, as resistant varieties are developed, this should be considered as well. Listed below are several varieties that have been tried in Georgia and have proven to be acceptable regarding these characteristics.
| Commercial Carrot Varieties for Georgia | |||||
| Variety | Source | Maturity | Use | Length | Tops |
| Varieties for full-season production | |||||
| Apache | SunSeeds | Medium | Fresh | 10" | mod strong |
| Choctaw | SunSeeds | Medium | Fresh/Proc | 10" | strong |
| Navajo | SunSeeds | Medium | Fresh | 9" | strong |
| Cheyenne | SunSeeds | Medium | Fresh | 8-10" | strong |
| Varieties for production only through mid-March | |||||
| Indiana | Bejo | Med-Late | Fresh/Proc | 10" | mod strong |
| Six Pak | Harris Moran | Early-Med | Fresh | 7-8" | strong |
| Topnotch (XPH 3918) | Asgrow | Medium | Fresh | 8-10" | mod strong |
| Vita Sweet 691 | Abbot & Cobb | Early | Fresh | 9-10" | strong |
Soil and Nutrition Management
Soil Requirements
Muck soils or loose, friable sandy loam soils are ideal for carrot production. Although heavier soils are not ideal, carrots can successfully be grown on heavy-textured soils under irrigated conditions. Short, blunt types are often grown on heavier soils. Sandy loam soils allow proper growth and development of a long, smooth, straight root. Soils cannot have excessive stones, pebbles and debris since this can cause forked or misshapen roots. Soils should also be well drained as carrots will not perform well under water-logged conditions. Sites should be selected that have loose, friable soils to a depth of 12-14 inches without pebbles. Deep sandy soils can also be used although they may require more frequent irrigation. Drain tiles should be installed on flatwood type soils that are subject to water-logged conditions. Preparing high beds to avoid wet conditions in these soils is not recommended since under drier circumstances these beds will dry out and cause damage to carrot tops and shoulders.
Land Preparation
Proper land preparation should begin by deep turning soils to bury any litter and debris and breaking soils to a depth of 12-14 inches. Compacted soils or those with tillage pans can benefit from subsoiling to break the compacted areas. If uncorrected, compact soil or tillage pans can result in restriction of root expansion. It is best to apply lime after deep turning to prevent turning up acid soil after lime application. After turning, mark beds using tractor tracks. Prepare a good seedbed using bed-shaping equipment. Do not use disks or rototiller to avoid soil compaction. Carrots in Georgia should be planted on a slightly raised bed (2-3 inches) to improve drainage. After beds are tilled and prepared for seeding, it is best to allow the beds to settle slightly before planting. Avoid other tillage practices that can increase soil compaction.
Following in the same tracks for all field operations will help reduce compaction in planting areas.
Fertilization and Nutrition Management
Carrot fertilization is one of the most critical components of successful production. Carrots have a medium requirement for nitrogen, however, timing of applications is just as critical as amount of fertilizer used. Carrots require a pH of between 6.0 and 6.5 for optimum growth. Acquire soil test recommendations several months prior to planting in order to make needed lime applications two to three months in advance of planting. If soil tests indicate magnesium is required use dolomitic lime.
Apply potassium and phosphorous according to soil test results. Recommendations for potassium and phosphorous based on various soil test indices are shown in Table 1. Apply one-third to one-half of potassium and all phosphorous banded in the width of the bed and incorporated prior to planting. The remaining potassium should be applied in two to three sidedress applications. Apply 1 pound of boron and 10 pounds of sulfur per acre. If soil test zinc is low, apply 5 pounds of actual zinc per acre.
Nitrogen requirements for carrots will be from 90 to 120 pounds per acre. It is imperative that nitrogen be applied in small quantities. These spoon fed applications allow the crop to better utilize nitrogen and helps to prevent splitting from growth spurts. Incorporate one-sixth to one-fourth of the recommended nitrogen into the bed prior to planting. Apply remaining nitrogen in four to six sidedress applications. Never apply more than 15 pounds of nitrogen per acre at any one time. Additional applications may be necessary if leaching rains occur. Since carrots may remain in the field from 110 to 130 days, nitrogen applications should be spread out over the length of the growing season by making applications every two to three weeks, depending on rainfall. Applications through pivot irrigation can be used when wet conditions prevent tractor operations. This can also reduce field traffic and thus compaction.
Calcium, boron and magnesium should be applied according to soil test recommendations. However, the use of foliar applications of these nutrients may be beneficial to maintain proper growth. Two to four applications of two quarts of CAB (or similar material) beginning with the first sidedressing and 3-5 pounds of MgSO4 along with the first sidedressing can be beneficial.
Although plant tissue analysis is a good tool to use in all vegetable crops, in carrots it is almost essential. Since winter weather conditions often make scheduled fertilizer applications difficult, the use of tissue analysis to keep track of nutritional status can help avoid problems before they become yield limiting. Proper levels of various nutrients at different stages of growth are shown in Table 2. See your county agent or crop consultant regarding proper techniques for tissue sampling and availability of laboratories that provide routine tissue analysis.
| Table 2. Plant tissue analysis standards by nutrient for carrots based on stage of growth. | |||||||||||
| Status | N | P | K | Ca | Mg | S | Fe | Mn | Zn | B | Cu |
| % | ppm | ||||||||||
| 0-60 days after seeding | |||||||||||
| Deficient | <1.8 | 0.2 | 2.0 | 2.0 | 0.2 | 0.2 | 30 | 30 | 20 | 20 | 4 |
| Adequate | 1.8-2.5 | 0.2-0.4 | 2.0-4.0 | 2.0-3.5 | 0.2-0.5 | 0.2-0.4 | 30-60 | 30-60 | 20-40 | 20-40 | 4-10 |
| High | >2.5 | 0.4 | 4.0 | 3.5 | 0.5 | 0.4 | 60 | 100 | 60 | 40 | 10 |
| > 60 days after seeding | |||||||||||
| Deficient | <1.5 | 0.18 | 1.0 | 1.0 | 0.25 | na | 20 | 30 | 20 | 20 | 4 |
| Adequate | 1.5-2.5 | 0.18-0.40 | 1.4-4.0 | 1.0-1.5 | 0.4-0.5 | na | 20-30 | 30-60 | 20-60 | 20-40 | 4-10 |
| High | >2.5 | 0.4 | 4.0 | 1.5 | 0.5 | na | 60 | 100 | 60 | 40 | 10 |
| Adapted from Carrot Production in Florida. Publication HS 722, a chapter of the Vegetable Production Guide for Florida, SP 170. Horticultural Sciences Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. July, 1995. | |||||||||||
Disease Management
Disease management in carrot production is necessary to produce high yields of high quality carrots. The major concern is the production of a disease-free and cosmetically clean carrot root. The main problems associated with carrot production are root-knot nematodes and diseases caused by fungi and bacteria. Carrots should be planted in well-drained, litter-free, deep turned soils. Certain crops like tobacco should be avoided because of the slow decomposition of the crop stubble. Also, nematode control in tobacco stubble is difficult to achieve because nematodes can withstand treatment as they are embedded in the root system and may not be exposed to the fumigant. Several root diseases will be discussed in this section.
Also, certain foliage diseases can affect carrots. In most cases, these never require any type of fungicide or bactericide application. However, under certain weather conditions, fungicide applications may be necessary. Basically, adequate fungicides and bactericides for managing the above ground diseases are available.
Root-Knot Nematode
 Figure 1. Root-knot galling.
Figure 1. Root-knot galling.By far, the most destructive problem is root-knot nematodes caused by Meloidogne sp. Root-knot nematodes are small eel-like worms that live in the soil and feed on plant roots. Since the root of the carrot is the harvested portion of the plant, no root-knot damage can be allowed. Root-knot causes poor growth and distorted or deformed root systems, which results in a non marketable root. Root-knot damage also allows entry for other diseases such as Fusarium, Pythium, and Erwinia. (Figure 1)
Soil assay is the best way to determine the numbers and kinds of nematodes to be dealt with in carrot production. A nematode assay should be made during the months of July, August, or September, depending on the current crop. Nematode populations are usually at a peak at about crop maturity. Thus, the most accurate numbers can be obtained during this period. If any root-knot nematodes are found, treatment is recommended. Good success has been obtained using field soil fumigation to eradicate root-knot nematodes in the root zone of carrots.
Soil-Borne Root Diseases
Depending on the cropping history of the field, Pythium, Southern Blight and Sclerotinia may cause problems. It is advisable to avoid fields where these diseases have been identified in the previous crop. Deep turning is also necessary to help prevent root diseases.
Pythium Blight
Pythium Blight is usually characterized by flagging foliage indicating some root damage is occurring. Under wet conditions, Pythium may cause serious problems to the root causing a white mycelium mat to grow on the infected area that rapidly turns to a watery soft rot. Forking of the root system is also a common symptom associated with Pythium infection. Pythium is probably the most isolated disease associated with carrot production in Georgia. Rotation is considered a major factor in reducing Pythium along with the use of fungicides. (Figures 2, 3)
 Figure 2. Root forking caused by Pythium. Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 2. Root forking caused by Pythium. Photo courtesy of J.O. Strandberg, Univ. Fla. Figure 3. Stumping caused by Pythium. Photo courtesy of J.O. Sandberg, Univ. Fla.
Figure 3. Stumping caused by Pythium. Photo courtesy of J.O. Sandberg, Univ. Fla.
Southern Blight
Southern blight is caused by the fungus Sclerotium rolfsii and can cause serious damage to carrots. This disease is usually associated with carrots remaining in the field after the soil begins to warm in the spring. This disease causes a yellow top to develop with a cottony white fungal growth associated with the upper part of the carrot root. The top of the root and the surrounding soil may be covered with a white mycelium with tan sclerotia developing as the disease progresses. Southern Blight is best controlled by using rotation and deep turning. (Figures 4, 5)
 Figure 4. Above ground symptoms and signs of infection caused by Schlerotium rolfsii. Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 4. Above ground symptoms and signs of infection caused by Schlerotium rolfsii. Photo courtesy of J.O. Strandberg, Univ. Fla.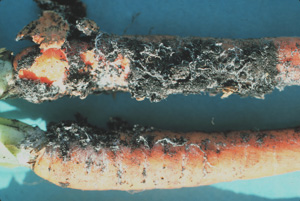 Figure 5. White mycelium of Schlerotium rolfsii on carrots. Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 5. White mycelium of Schlerotium rolfsii on carrots. Photo courtesy of J.O. Strandberg, Univ. Fla.
Sclerotinia Blight
Sclerotinia blight is caused by the fungus Sclerotinia sclerotiorum and can cause serious damage to the roots of carrots. This disease is usually worse under wet soil conditions. This is the same disease that causes the familiar raisin head in cabbage. White mycelium forms around the infected area and, later, dark sclerotia develop on the white mycelium, which is a good indicator of Sclerotinia rot. This disease causes a progressive watery soft rot of the carrot root tissue and is considered a potential problem in the production of carrots. Rotation and deep turning of the soil are recommended to reduce losses to this disease.
Rhizoctonia
This disease is caused by Rhizoctonia species and is associated with the carrot root. It causes brown to black lesions to develop on the sides of the carrot root. The disease is much worse under cool, wet conditions. Saturated soil conditions often enhance all soil-borne diseases, which are potential problems in carrot production. Rhizoctonia damage can be minimized by using rotation and good cultural practices. Soil fumigation will prevent damage with any of the soil inhabiting fungi, however soil fumigation is expensive and cultural practices may be used to reduce the injury without the expense of fumigation. (Figure 6)
 Figure 6. Symptoms caused by Rhizoctonia. Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 6. Symptoms caused by Rhizoctonia. Photo courtesy of J.O. Strandberg, Univ. Fla.Foiliar Diseases
Bacterial Blight
Bacterial Blight caused by the bacterium Xanthomonas carotae causes irregular brown spots on the leaves and dark brown streaks on the petioles and stems. The lesions on the foliage begin as small yellow areas with the centers becoming dry and brittle, with an irregular halo. The bacterium affects the leaflets, stems and petioles as the disease progresses. Some of these lesions may crack open and ooze the bacteria. These bacteria may be washed down to the crown of the plant causing brown lesions on the top of the root. The earlier the infection the more damage to the root. The bacterium is spread by splashing water and takes about 10-12 days before symptoms appear after inoculation. The bacterium progresses rapidly between 77 degrees and 86 degrees F. Rotation is a major factor in controlling Bacterial Blight. (Figures 7, 8)
 Figure 7. Foliar symptoms caused by Xanthomonas carota. Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 7. Foliar symptoms caused by Xanthomonas carota. Photo courtesy of J.O. Strandberg, Univ. Fla. Figure 8. A comparison of symptoms caused by Alternaria dauci (right) and Xanthomonas carota (left). Photo courtesy of J.O. Strandberg, Univ. Fla.
Figure 8. A comparison of symptoms caused by Alternaria dauci (right) and Xanthomonas carota (left). Photo courtesy of J.O. Strandberg, Univ. Fla.
Alternaria Blight
Alternaria blight is caused by the fungus Alternaria dauci. This disease causes small dark brown to black spots with yellow edges forming mostly on the leaf margins. The spot increases as the disease progresses and in some cases entire leaflets may be killed. In moist weather, the disease can move so rapidly it resembles frost injury. Such conditions can reduce the efficiency of mechanical harvesters which require strong healthy tops to remove the carrot from the soil. Alternaria may also cause damping off of seedlings and a black decay of roots. The spores and mycelium are spread by splashing rains, contaminated soil, or on cultivation tools. The disease can manifest itself in about 10 days after infection. The optimum temperature for Alternaria blight is 82 degrees F. (Figure 9)
 Figure 9. Alternaria dauci on seedling carrots. Photo courtesy of J.O. Sandberg, Univ. Fla.
Figure 9. Alternaria dauci on seedling carrots. Photo courtesy of J.O. Sandberg, Univ. Fla.Cercospora Leaf Blight
Cercospora blight is caused by the fungus Cercosproa carotae. This disease causes lesions to form on the leaves, petioles and stems of the carrot plant. The symptoms appear to mimic that of Alternaria blight but can be separated using a compound microscope. Cercosproa blight progresses in warm, wet weather and spots appear in about 10 days after infection. The youngest leaves are usually more susceptible to Cercosproa infection.
Insects
Carrots are subject to attack by numerous insect pests. Fortunately, in Georgia, many pest populations are not present or do not develop to economically damaging levels during the major production window from fall to spring. Soil insects, if present, may damage the roots and should be controlled with cultural practices and incorporation of soil insecticide. Some fumigants may provide some control of soil insects. Other pests that primarily attack the foliage can be monitored and treated as needed with foliar insecticides.
Soil Insects
Wireworms, mostly Conoderus spp., white grubs, Phyllophagus spp., and the granulate cutworm, Feltia subterranea, may be partially controlled with good cultural practices. Soil should be deep turned in sufficient time prior to planting to allow destruction of previous crop residue that may harbor soil insects. When possible, avoid planting just after crops slow to decompose, such as tobacco and corn. Avoid planting behind peanuts and root crops such as sweet potatoes and turnips. If a field has a history of soil insect problems, either avoid these or broadcast incorporate a soil insecticide prior to planting. Plantings in fields recently in permanent pasture should be avoided as should fields recently planted to sod/turf, although these are not as critical. Fields with a history of whitefringed beetle larvae, Graphognathus spp., should not be planted to carrots because there are no currently registered insecticides effective on this pest. (Figures 10, 11, 12, 13)
 Figure 10.
Figure 10.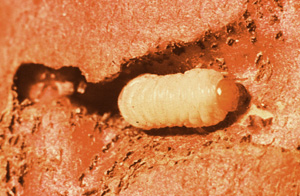 Figure 11.
Figure 11. Figure 12.
Figure 12.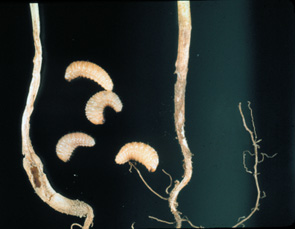 Figure 13.
Figure 13.
Flea beetle larvae, Systena spp., can damage roots by feeding on the surface too the cortex. The damage will take on the appearance of narrow s-shaped canals on the surface. Flea beetle larvae can be prevented easily with soil insecticides.
The seedcorn maggot, Hylemya platura, is an opportunistic pest that takes advantage of crops that are under stress or where there is decaying organic matter. Plants may be considered under stress for several reasons such as freeze damage, nutritional deficiencies, herbicide injury, drought, wind and sand injury and diseases to mention a few. At-planting soil insecticides will prevent the development of maggot infestations for several weeks after planting, however, as residues decrease late season infestations may develop, especially from late January through April. Seedcorn maggots cannot be effectively controlled after the infestation begins. If plants become stressed during the period of high root maggot potential, preventive applications of insecticides should be sprayed every seven days until the stress is minimized. (Figure 14)
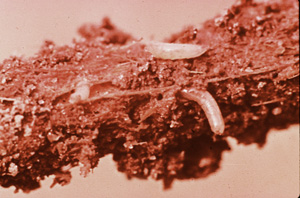 Figure 14.
Figure 14.Foliar Insects
Foliar insect pests may be monitored and insecticides applied as needed. Very often carrots may be grown without the addition of any foliar insecticides. Carrots should be scouted at least once per week for developing populations of foliage pests.
Aphids
Several species of aphids may develop on carrots. The most common aphids to inhabit carrots in Georgia are the green peach aphid, Myzus persicae and the cotton or melon aphid, Aphis gossypii. Both of these aphids may infest carrots in the fall after other cultivated hosts, such as cotton and vegetables, are harvested. There are no treatment thresholds for making control decisions. Often parasitic wasps and fungal diseases will control these aphids. If populations persist and colonize plants rapidly over several weeks and honeydew or sooty mold is observed readily, then foliar insecticides are justified.
Flea Beetles
Fleas beetle adults, Systena spp., may cause severe damage to the foliage on occasion. If carrots are attacked during the seedling stage and infestations persist over time, an insecticide application may be necessary. Minor feeding is no cause for alarm. If plants are in the cotyledon to first true leaf stage, treatments should be made if damage or flea beetles are observed on more than 5 per-cent of the plants. After plants are well established, flea beetles should be controlled only if foliage losses are projected to be moderate to high, e.g., 15 percent or more.
Vegetable Weevil
The adult and larvae of the vegetable weevil, Listroderes difficilis, may attack carrots from late December through April. They are most prevalent from late January through early April. The adult and larvae feed on the foliage. Vegetable weevil larvae often will feed near the crown of plants and, if shoulders are exposed at the soil surface, larvae will feed on tender carrots. Treatments are justified if adults or larvae and damage are easily found in several locations. [Figures 15 (larvae),16 (adult)]
 Figure 15.
Figure 15. Figure 16.
Figure 16.
Armyworms
The armyworm, Pseudaletia unipuncta, can cause damage from late winter through spring. Armyworms may move from grain crops or weeds into carrots or adults may lay eggs directly on carrot plants. Armyworms are easily controlled with foliar insecticides. There are no action thresholds but, if foliage is excessive, an insecticide application may not be justified. (Fig. 17)
 Figure 17.
Figure 17.Beet Armyworm
The beet armyworm, Spodoptera exigua, infests carrots in the late spring. Usually natural predators and especially parasites regulate beet armyworm populations below economically damaging levels.
Whiteflies
The silverleaf whitefly, Bemesia argentifolii, may be a problem from August through October during the early seedling stage of fall plantings. Silverleaf whitefly migrate from agronomic crops and other vegetables during the late summer. Infestation may become severe on carrots grown in these production areas. Often whiteflies may be controlled by several natural enemies and diseases by early fall so, treatments may not be justified. However, if whiteflies develop generally heavy populations, treatment on young plantings is justified. (Fig. 18)
 Figure 18.
Figure 18.
Managing Weeds in Carrots
Weed control in carrots is an essential component of producing a high-yielding and high-quality crop. Weeds compete with carrots for light, water, nutrients, and physical space. In addition, many weeds may impair the harvesting process, either through improper digging or contamination of the harvested crop. Weeds can also harbor deleterious insects and diseases. The presence of plant residue from weeds or other plants during fumigation can also decrease the effectiveness of most soil fumigation materials, thus increasing the problems associated with nematodes.
Most of the fumigants used in carrots grown in Georgia will not provide adequate weed control, therefore additional weed control measures must be employed. Carrot growers face a multitude of weed problems including both summer and winter annual broadleaf weeds and grasses. These may include sicklepod (Cassia obtusifolia), crabgrass (Digitaria spp.), pigweeds (Amaranthus spp.), carpetweed (Mollugo verticillata), lambsquarters (Chenopodium album), wild radish (Raphanus raphinistrum), cut-leaf evening primrose (Oenthera lanceolata), and several others.
Prevention is the first step in managing weeds in carrots. Avoid areas that contain heavy infestations of perennial weeds such as bermudagrass and nutsedge. Proper tillage, insect and disease control and fertility will help to ensure a healthy crop. Planting density will also allow the carrots to out-compete many weeds. Mechanical cultivation is generally not used in carrot production due to the tight row spacing (several rows on a bed). However, mechanical cultivations will help control weeds between rows particularly during early fall planting.
Chemical weed control is often used in carrot production and recommendations may be found in the most current Georgia Pest Management Handbook. Chemical weed control in carrots relies heavily on the herbicide linuron (Lorox, others). Linuron provides excellent control of most weeds found in carrots and is used postemergence over-the-top. Although good control of large weeds can be achieved with this product, it is critical to eliminate the weeds at an early stage to reduce the deleterious effects of competition. The lower rate should be used on smaller carrots (tops <2-4 inches) and during warmer weather. A crop oil may be added to improve control of larger weeds, although this may increase the chance of crop injury. The best rule of thumb is to avoid the use of crop oil on smaller carrots.
Sencor (metribuzin) can also be used for post-emer-gence weed control but certain varieties may be injured by this herbicide. Due to the limited use of this herbicide in Georgia a list of sensitive varieties has not been developed to date (consult the Georgia Pest Management Handbook). Trifluralin can also be used as a pre-plant incorporated treatment for the control of most annual grasses and several small-seeded broadleaf weeds. This herbicide should be applied before planting and soil incorporated to a depth of 2-3 inches. Trifluralin may provide early season weed suppression, allowing the carrots to emerge and begin seedling growth in the absence of heavy weed com-petition. Fluazifopmethyl (Fusilade) can be used for post-emergence weed control and provides good control of most annual and perennial grasses.
One of the worst weeds in carrots, as in most crops, is nutsedge. Most infestations are limited to yellow nutsedge (Cyperus esculentus), which is found throughout Georgia. Linuron and metribuzin may provide some temporary suppression, while trifluralin and fluazifop have no effect. Nutsedge causes the greatest problem in the early fall, during carrot emergence and seedling growth. This limits the use of higher rates of linuron for some suppression due to the small carrot size. Unfortunately, the lower rates give no control. The stale-seedbed technique may provide some control and can be effectively used in carrots for nutsedge because nutsedge will emerge several days before the carrots. A non-selective material such as Roundup Ultra or Gramoxone Extra is commonly used. As with most weed management strategies, proper weed identification, site selection and timing are the critical issues in controlling weeds in carrots.
Irrigation Management for Carrots
Carrots are generally shallow rooted (4 to 6 inches). However, in Georgia soils the effective rooting depth and actual rooting depth will vary considerably, depending on soil conditions and cultural practices. The restricted rooting depth and the fact that carrots are commonly grown in sandy soils with a low water holding capacity make irrigation necessary for consistently high yields of quality carrots in Georgia.
The most serious yield reductions result from water stress during germination and during root expansion. Water deficits during the establishment of carrots delay maturity and may cause gaps in production. Water stress in the early vegetative stage results in reduced leaf area and reduced yield.
Several types of irrigation systems may be used successfully on carrots in the Southeast. Ultimately, the decision as to which type to choose will be based on one or more of the following factors:
- Availability of existing equipment.
- Field shape and size.
- Amount and quality of water available.
- Labor requirements.
- Fuel requirements.
- Cost.
Sprinkler Irrigation
Currently, most carrots are irrigated with some type of sprinkler irrigation. These systems include center pivot, linear move, traveling big-gun, permanent set and portable aluminum pipe with sprinklers. Any of these systems are satisfactory, if they are used correctly. There are, however, significant differences in initial cost and labor requirements.
Any sprinkler system used on carrots should be capable of delivering at least an inch of water every four days. In addition, the system should apply the water slowly enough to prevent run-off. With most soils, a rate less than two inches per hour safely prevents runoff.
Sprinkler systems with a high application uniformity (center pivot and linear move) can be used to apply fertilizer through the system. This increases the efficiency of fertilizer utilization by making it readily available to the plant and reducing leaching. With soils containing clay particles, high volume, large droplet sprinklers can cause stand reduction.
Scheduling Irrigation
The water used by a crop and evaporated from the soil is called evapotranspiration (ET). Factors that affect ET are stage of crop growth, temperature, relative humidity, solar radiation, wind and plant spacing.
The following are general recommendations for irrigation rates on sprinkler irrigated carrots:
- Make sure good soil moisture exists before and after seeding. Apply 1/10 to 1/4 inch water whenever soil in top two inches becomes dry (about two to three times daily when weather is dry).
- After plants are established apply 3/4 inch every five days during dry weather.
- If wilting occurs before noon, increase frequency of irrigation.
- Sandy soils may require more frequent, lighter applications than heavier soils to prevent moisture stress.
Soil moisture monitoring can be used to fine tune irrigation applications. This ensures that soil moisture is adequate to prevent crop stress. The irrigation schedule should be adjusted whenever soil moisture measurements indicate overly wet or dry conditions.
Soil moisture may be monitored using either electric resistance blocks (such as the Watermark™ soil moisture sensor) or tensiometers. Install two sensors at each monitoring location -- one about 4-6 inches deep and one about 12 inches deep. Each field should have a minimum of two monitoring locations -- more for fields larger than 20 acres or if soil types vary considerably.
The 4-6 inch sensors are near the middle of the root zone and will indicate when irrigation should be started. Up until tops are fully expanded, readings should not exceed 50 centibars. Afterward, they should not exceed 30 centibars. The optimum range for soil moisture is 5-30 centibars. The 12-inch sensor is used to evaluate previous irrigations. If readings remain low (less than five centibars), then irrigation amounts should be decreased. If they continue to increase even after an irrigation, then irrigation amounts should be increased. Read soil moisture sensors at least three times per week during dry weather.
Pesticide Application
Two types of sprayers, boom and air-assisted, are used for applying insecticides, fungicides, herbicides and foliar fertilizers. Air-assisted sprayers (Figure 19) utilize a conventional hydraulic nozzle and air is used to force the spray into the plant foliage. Boom sprayers (Figure 20) get their name from the arrangement of the conduit that carries the spray liquid to the nozzles. Booms or long arms on the sprayer extend across a given width to cover a swath as the sprayer passes over the field.
 Figure 19.
Figure 19. Figure 20.
Figure 20.
Pumps
Consider three factors in selecting the proper pump for a sprayer:
- Capacity. The pump should be of proper capacity or size to supply the boom output and to provide for agitation (5 to 7 gallons per minute (gpm) per 100-gallon tank capacity). Boom output will vary, depending on the number and size of nozzles. Also, 20 to 30 percent should be allowed for pump wear when determining pump capacity. Pump capacities are given in gallons per minute.
- Pressure. The pump must produce desired operating pressure for the spraying job to be done. Pressures are indicated as pounds per square inch (psi).
- Resistance to corrosion and wear. The pump must be able to withstand the chemical spray materials without excessive corrosion or wear. Use care in selecting a pump if wettable powders are to be used as these materials will cause pump wear.
Before selecting a pump, consider cost, service, operating speeds, flow rate, pressure and wear. For vegetable crops, a diaphragm pump is preferred because of serviceability and pressures required.
Nozzles
Nozzle selection is one of the most important decisions to be made related to pesticide applications. The type of nozzle determines not only the amount of spray applied, but also the uniformity of the applied spray, the coverage obtained on the sprayed surfaces, and the amount of drift that can occur. Each nozzle type has specific characteristics and capabilities and is designed for use under certain application conditions. The types which are commonly used for ground application of agricultural chemicals for carrots are the fan and cone nozzles.
Herbicides
The type of nozzle used for applying herbicides is one that develops a large droplet and has no drift. The nozzles used for broadcast applications include the extended range flat fan, drift reduction flat fan, turbo flat fan, flooding fan, turbo flooding fan, turbo drop flat fan, and wide angle cone nozzles. Operating pressures should be 20 to 30 psi for all except drift reduction and turbo drop flat fans, flooding and wide angle cones. Spray pressure more than 40 psi will create significant drift with flat fans nozzles.
Drift reduction and turbo drop nozzles should be operated at 40 psi. Flooding fan and wide angle cone nozzles should be operated at 15 to 18 psi. These nozzles will achieve uniform application of the chemical if they are uniformly spaced along the boom. Flat fan nozzles should overlap 50 to 60 percent.
Insecticides and Fungicides
Hollow cone nozzles are used primarily for plant foliage penetration for effective insect and disease control, and when drift is not a major concern. At pressures of 60 to 200 psi, these nozzles produce small droplets that penetrate plant canopies and cover the underside of the leaves more effectively than any other nozzle type. The hollow cone nozzles produce a cone shaped pattern with the spray concentrated in a ring around the outer edge of the pattern. Even fan and hollow cone nozzles can be used for banding insecticide or fungicides over the row.
Nozzle Material
Various types of nozzle bodies and caps, including color coded versions and multiple nozzle bodies, are available. Nozzle tips are interchangeable and are available in a wide variety of materials, including hardened stainless steel, stainless steel, brass, ceramic, and various types of plastic. Hardened stainless steel and ceramic are the most wear-resistant. Stainless steel tips, with corrosive or abrasive materials, have excellent wear resistance. Plastic tips resist corrosion and abrasion and are proving to be very economical for applying pesticides. Brass tips have been common but wear rapidly when used to apply abrasive materials such as wettable powders. Brass tips are economical for limited use, but consider other types for more extensive use.
Water Rates (GPA)
The grower who plans to use spray materials at the low water rates should follow all recommendations carefully. Use product label recommendations on water rates to achieve optimal performance. Plant size and condition influence the water rate applied per acre. Examination of the crop behind the sprayer before the spray dries will give a good indication of coverage.
Agitation
Most materials applied by a sprayer are in a mixture or suspension. Uniform application requires a homogeneous solution provided by proper agitation (mixing). The agitation may be produced by jet agitators, volume boosters (sometimes referred to as hydraulic agitators), and mechanical agitators. These can be bought separately and installed on sprayers. Continuous agitation is needed when applying pesticides that tend to settle out, even when moving from field to field or when stopping for a few minutes.
Nozzle Arrangements
When applying insecticides and fungicides, use a broadcast boom arrangement. Place nozzles on 10 to 12 inch centers for complete coverage of the plant.
Calibration
The procedure below is based on spraying 1/128 of an acre per nozzle or row spacing and collecting the spray that would be released during the time it takes to spray the area. Because there are 128 ounces of liquid in 1 gallon, this convenient relationship results in ounces of liquid collected being directly equal to the application rate in gallons per acre.
Calibrate with clean water when applying toxic pesticides mixed with large volumes of water. Check uniformity of nozzle output across the boom. Collect from each for a known time period. Each nozzle should be with-in 10 percent of the average output. Replace with new nozzles if necessary. When applying materials that are appreciably different from water in weight or flow characteristics, such as fertilizer solutions, etc., calibrate with the material to be applied. Exercise extreme care and use protective equipment when active ingredient is involved.
- From Table 3, determine the distance to drive in the field (two or more runs suggested). For broadcast spraying measure the distance between nozzles. For band spraying, used band width. For over the row or directed use row spacing.
- Measure the time (seconds) to drive the required distance; with all equipment attached and operating. Maintain this throttle setting!
- With sprayer sitting still and operating at same throttle setting or engine RPM as used in Step 2, adjust pressure to the desired setting. Machine must be operated at same pressure used for calibration.
- For broadcast application, collect spray from one nozzle or outlet for the number of seconds required to travel the calibration distance.
For band application, collect spray from all nozzles or outlets used on one band width for the number of seconds required to travel the calibration distance.
For row application, collect spray from all outlets (nozzles, etc.) used for one row for the number of seconds required to travel the calibration distance. - Measure the amount of liquid collected in fluid ounces. The number of ounces collected is the gallons per acre rate on the coverage basis indicated. For example, if you collect 18 ounces, the sprayer will apply 18 gallons per acre. Adjust applicator speed, pressure, nozzle size, etc. to obtain recommended rate. If speed is adjusted, start at Step 2 and recalibrate. If pressure or nozzles are changed, start at Step 3 and recalibrate.
| Table 3. Distance to measure to spray 1/128 acre. One ounce discharged equals one gallon per acre. | |||
| Nozzle Spacing (inches) | Distance (feet) | Nozzle Spacing (inches) | Distance (feet) |
| 6 | 681 | 20 | 204 |
| 8 | 510 | 22 | 186 |
| 10 | 408 | 24 | 170 |
| 12 | 340 | 30 | 136 |
| 14 | 292 | 36 | 113 |
| 16 | 255 | 38 | 107 |
| 18 | 227 | 40 | 102 |
| To determine a calibration distance for an unlisted spacing, divide the spacing expressed in feet into 340. Example: Calibration distance for a 13" band = 340÷13/12=313 feet. | |||
Fumigation Equipment
Fumigants are applied as broadcast and as row treatments. Row treatment is used most often on cotton, tobacco, peanuts and vegetables because of the economy of this type treatment. Machines for applying broadcast and row treatments are quite similar. The major difference is in the spacing of applicator injectors or other placement devices.
Broadcast Treatment
For broadcast treatment, applicator devices are usually set to inject parallel streams of fumigant 10 to 12 inches apart at a depth of 10 to 12 inches or deeper. No consideration is given to where crop rows will be located or in what direction applicators are operated relative to row direction.
Applicator injectors can be attached to cultivator frames in place of a cultivation tool, to tool bars or bed shapers. They are mounted in a staggered pattern, with alternate injectors on a second line several inches behind the front line. This reduces problems caused by trash accumulating on injectors or by rocks and other material bridging the space between injectors.
Either chisel, subsoiler, conventional knife or rolling knife type injectors can be used. The rolling knife-type injector will cut through some trash that may accumulate on the chisel type. Figure 21 shows a combination colter and subsoiler shank broadcast fumigant applicator.
Furrows opened by injectors must be closed. Under some conditions, soil movement behind the injectors will close furrows. Most of the time, however, covering knives or discs, a press wheel or some drag-type smoothing device (broad or roller) should be attached to the applicator machine to close injector furrows. If furrows are closed by a separate operation, it should follow directly behind the applicator machine so that furrows will be closed immediately -- a matter of seconds.
Broadcast treatment can also be applied with a moldboard (Figure 22) plow during land preparation. Chemical tubes are attached to the plow to deliver the fumigant to the bottom of plow furrows immediately ahead of each plow bottom. Thus, the fumigant is discharged and covered immediately.
 Figure 21.
Figure 21. Figure 22.
Figure 22.In-Row Treatment
One or two injectors are used per row, with one injector being used most often. When a single knife or chisel is used, mount it to where the fumigant is under-neath the position of the crop row. When treating during planting operations, mount applicators 1 to 2 inches to one side of the row to reduce interference with seed placement and to ensure that the seed does not fall into fumigant furrow.
When two injectors are used per row, they are spaced 10 to 12 inches apart and positioned so the row will be centered between them.
In both one and two injector row treatments, the applicators are operated so the fumigant is placed approximately 10 to 12 inches below soil surface.
When row treatments are applied in advance, of planting, the position of fumigant placement must be marked so that seed will be planted in the treated area. The treated area will be 16 to 20 inches wide for a single injector and 18 to 24 inches wide for two injector applications. If crop plants are not placed in the treated area, no benefit will be gained.
A common practice is to form beds over injector furrows at the time of application. This marks the location of the treated area and closes injector furrows to seal volatile fumigants in the soil.
Equipment
Application systems can be either PTO, electric or ground driven pumps. A PTO or electric pump draws fumigant from the tank and pumps it into the distribution manifold. Excess fumigant is diverted back to the tank through a bypass valve. Flow rate is regulated by metering discs located shank outlets and by the line pressure (Do not exceed 25 psi.), which is regulated by the bypass valve adjustment. When the flow to the shanks is stopped, all of the chemical is bypassed to the tank. A check ball screen should be placed between the hose and the metal tube to the injection shank. This will minimize end row drips and prevent clogging of the orifice plate.
Compatible Materials
Some materials may have a violent reaction when com-ing in contact with a particular soil fumigant. Listed are some materials compatible and not compatible with fumigants.
Compatible: HD polyethylene, nylon, Teflon, Viton, stainless steel, mild steel, brass, copper, black iron and cross-linked polyethylene.
Not Compatible: polypropylene, rubber, plastic, aluminum, magnesium, zinc, cadmium, galvanized steel, fiberglass, EPDM, Nuna-N, neoprene and PVC.
Calibration
Always calibrate with clean water because fumigants are very corrosive. Fumigant applicators can be calibrated using the procedure for a conventional sprayer (previous section). Make certain to covert fumigant rates to water rates prior to the calibration.
The conversion factor for Telone II is 1.1. Telone C-17 has a conversion factor of 1.13. Water rate = Fumigant rate multiplied by conversion factor.
If application rates are given in rate per 100 feet of row, simply mark off 100 feet and measure the time to travel with equipment in operation that distance. Next, with equipment in a stationary position, collect in ounces flow from the outlet(s) for each row for the time to travel the 100 feet. Adjust the desired rate per 100 feet of row by multiplying by the conversion factor. If the amount collected is different from the desired rate, either change orifice plate or pres-sure setting.
Maintaining Equipment
Always conduct periodic checks of the systems for leaks. Each year make a practice to replace hose lines and tanks seals. Check valves, strainers, orifices and pressure regulators should be cleaned and in working order.
Cleanup and maintenance programs are much more important when fumigants are used than when less corrosive chemicals are used, even when sprayer components are made of the more corrosive resistant material. Diesel fuel or kerosene is a good material to use for cleaning machinery after applying fumigants. After fumigant applications, always flush your system with diesel fuel and fill pump with new motor oil.
Good Agricultural Practices In the Harvest Handling and Packaging Of Fresh Carrots
Introduction
When newspaper headlines and radio or television newscasts announce that a foodborne illness associated with fresh produce has occurred, the entire industry from grower to retailer should take serious notice. Today's produce is increasingly being put under the microscope as a potential carrier of safety hazards. Carrot growers and shippers are urged to take a proactive role in minimizing the food safety risks for their crops.
Quality and Safety
Carrot quality and safety are often perceived by consumers to mean the same thing. Good quality carrots may be visually appealing and delicious, yet may contain pathogens or toxins that can cause illness to the consumer. Safe product, in contrast, may be discolored, over mature and unappealing, yet present no hazard to the consumer. Unfortunately, the safety of fresh carrots cannot be determined by its outward appearance or condition.
Field Sanitation Program
Raw Product Safety
Ensuring raw product safety begins with preventing hazards in the carrot field. The best guarantee of a safe raw product is a proactive Food Safety program designed and implemented to identify and prevent hazards during fresh carrot production and postharvest handling. Growers/shippers should familiarize themselves with safe production practices so they might be viewed as qualified suppliers among potential buyers. Some issues of concern during carrot production are summarized in Table 4.
| Table 4. Potential hazards during carrot production | |||
| Production Factor | Potential Hazard | Prevention | Documentation |
| Land Use | Fecal contamination (source of pathogens) from animals | No grazing animals or feed-lots on/near production land | Grower certification of no recent animal husbandry on land used |
| Toxic pesticide residues in soil | Review pesticide history for plant back restrictions | Pesticide selection/application records | |
| Fertilizers | Pathogenic bacteria from organic fertilizers | Use inorganic fertilizer | Credible test results |
| Heavy metal toxicity from sewage sludge | Use certified organic fertilizers or tested and approved sludge | Credible test results | |
| Irrigation Water | Pathogenic bacteria from surface water | Test/monitor water supply | Water test results |
| Heavy metal/pesticide residues in groundwater | Test/monitor water supply | Water test results | |
| Pesticide Use | Illegal/hazardous residues on product | Employ only professional, licensed applicators and monitor pesticide use | Examine applicator records; test for residues if contamination suspected |
| Hand Harvesting | Fecal contamination of product | Field worker personal hygiene; field washing/sanitizing facilities available | Training programs on worker hygiene |
| Field Containers | Soil and human pathogens | Use plastic bins; clean/sanitize all containers | Field sanitation records |
Land Use History
Grazing animals on or near crop land can introduce pathogenic (to humans) bacteria to the soil. Growers should ensure that land has not been used for animal husbandry and that it is not in proximity to animal feedlots or water runoff from grazing lands. Past improper use of pesticides can result in hazardous residues on raw product. Buyers might insist on letters of guarantee from grower/ shippers that the land is suitable and safe for the crops being produced. Growers should be aware of the field history regarding pesticides and any previous uses that might have introduced heavy metals into the soil.
Fertilizer Use
Insufficiently composted organic fertilizers may contain pathogenic (to humans) bacteria derived from animal or human feces. If organic fertilizers are used, be sure that they have been sufficiently composted so pathogens are not present. Inorganic fertilizers originate from generally, non-toxic, synthetic chemicals, so no pathogens are present. Composted sewage sludge should not be used as it may contain pathogens as well as heavy metal contamination.
Irrigation
Natural surface water (e.g., canal, lake, pond) may provide enough organic matter to support the growth of bacterial pathogens. Surface water may be used but should be tested for the presence of the pathogenic bacterium Escherichia coli (E. coli), which is an indicator of fecal contamination. An infectious dose may be as low as 10 organisms per milliliter. Groundwater is less likely to harbor human pathogens but should be analyzed for heavy metal and pesticide contamination.
Overhead irrigation is more likely to spread contamination to above-ground plant parts than is root-zone irrigation. Growers must be able to document answers to the following questions: Are irrigation practices safe? What is the water source? How is water stored? Are animals being raised nearby? What tests are performed to ensure the purity and safety of the water?
Pesticide Usage
Inspection, monitoring and documentation of proper use of pesticides will prevent unsafe or illegal pesticide residues from contaminating the raw product. Growers must be able to answer the following questions: Do you oversee your pesticide-spraying program? Do you have record-keeping procedures to track all spraying of this crop? Do you or the state/federal government regularly test your crops for residue levels?
Harvesting
Mechanical harvesting can wound produce, encouraging contamination from the soil. Hand harvesting may lead to pathogen contamination if field workers practice poor hygiene. Field crews must be trained and monitored for personal hygiene (hand washing activities), and portable bathroom and hand-washing facilities must be provided in the field.
Field Containers (boxes, buckets, bins, etc.)
Containers for harvesting fresh produce should be non-toxic, easy to clean and free of extraneous materials (e.g., nails, wood splinters, etc.) that can carry over into processing. They must be approved by the U.S. Department of Agriculture (USDA) or the Food and Drug Administration (FDA) for field use. After detergent cleaning, field bins, buckets, etc., can be sanitized using a very strong sodium hypochlorite solution dispensed from a high-pressure sprayer.
Post Harvest Handling
Harvest Quality
Fresh Georgia carrots should be harvested before reaching full maturity. These carrots average 7 to 9 inches in length, 1.5 inches in diameter, and have a mild, sweet flavor and a bright external color. All Georgia carrots are machine harvested.
Harvesting and Packaging
Machine harvested carrots are loosened under the row by pull-like devices and elevated out of the soil onto belts that grasp the carrot tops. These tops are cut mechanically and allowed to fall back into the field while the trimmed roots are elevated to trucks for bulk transport to the packing house. At the packing house, carrots are off loaded into a dry dump tank, conveyed through a cylindrical revolving cleaner to remove dirt and sand, and then they enter a revolving drum washer with spray nozzles. Washed carrots then proceed through a series of sizing belts, where they are sorted by length and diameter. Diverging rollers are used to separate roots for diameter, and an inclined, vibrating platform with graduated hole sizes separates roots according to length. Sized carrots drop onto conveyor belts for hand grading to remove culls and are then hand packed into one-, two-, three- and five-pound polyethylene bags, which are subsequently palletized into 50 pound, heavy polyethylene master bags.
Mechanical damage to the roots can be minimized by the proper operation of all harvesting, sizing and grading equipment. Excessive speed or overloading of equipment will result in an increase of mechanical damage to the carrots. Such bruising or cutting will open up avenues for greater water loss and invasion of decay microorganisms.
Precooling and Storage Requirements
Carrots have a high rate of respiration, which means field heat must be removed before dense packaging and storage. Precooling is accomplished by using cold water in the revolving drum washer or running field bins through a hydrocooler. Keep temperatures at 40 degrees F.
Topped fresh market carrots are very perishable and rapidly transpire moisture from even trimmed roots. Wilting symptoms become evident with as little as a 3 percent weight loss. Therefore, high humidity (95-100%) is imperative in storage. Free moisture, if allowed to condense on carrots will promote decay. Good air movement is necessary to prevent decay during storage. Topped carrots should be held at 32 degrees F. If the above conditions are met, the shelf life of immature, topped carrots is four to six weeks.
Quality Defects
One of the most common signs of visual quality loss is a lack of firmness: Carrots become limp and soft, flabby, and/or shriveled. Other quality defects causing carrots to be downgraded by the buyer or due to a USDA inspection include the following: Non-uniform shape, growth cracks, insect damage, poor color, roughness, green core, sunburn injury, poor trimming, evidence of freeze damage and decay.
Mixed Load/Storage Compatibility
Carrots have the same storage requirements as beets, broccoli, brussels sprouts, cabbage, cauliflower, celery, collards, sweet corn, endive, greens, lettuce, green onions, parsley, parsnips, peas, radishes, snow peas, spinach, and watercress. Therefore, these crops can be stored together without deleterious effects. However, carrots should not be stored with ethylene producing crops (e.g., cantaloupes, apples, etc.). Carrots are sensitive to as little as 0.5 ppm ethylene. This will impart a perceptible bitter flavor to the carrots.
Postharvest Decay
Postharvest decay organisms are obligate parasites and therefore, do not normally enter the product through healthy, exterior tissue. These organisms require mechanical damage or weakening of tissue before they can enter. Spoilage losses can be minimized if the following precautions are observed: Use new or disinfected storage containers, handle carrots carefully to prevent injuries, precool roots properly, and maintain carrots at a constant temperature slightly above 32 degrees F.
The most important decays of carrots in storage are gray mold rot (Botrytis), watery soft rot (Sclerotinia), crater rot (Rhizoctonia), fusarium rot, rhizopus soft rot, bacterial soft rot, black rot (Stemphylium), and sour rot (Geotrichuim).
Sanitary Guidelines for Packinghouse Operations
Receiving Incoming Product
Harvest crews should remove as much dirt and mud from the product as is possible before the product leaves the field. An area should be set aside in the receiving yard so pallets can be cleaned before dumping in bins or cooling.
Water Sanitation
Water used in cleaning and cooling should be chlorinated at a concentration of 75 to 100 ppm of free chlorine. Chlorination can be accomplished using a gas injection system, adding bleach or using calcium hypochlorate tablets. Chlorination levels in the water should be monitored frequently during operation, through the use of chlorine litmus paper, or more accurately with a chlorine test kit. Water pH should be maintained between pH = 6.5 - 7.5 to avoid having to use excess chlorine in order to maintain recommended free chlorine levels. Excessive use of chlorine causes gassing off (objectionable chlorine odor -- gas can irritate worker's skin, is corrosive to equipment and increases sanitation cost).
Employee Hygiene
Good employee hygiene is very important. Employee training, health screening and constant monitoring of packinghouse sanitation practices (hand washing, personal hygiene) are important in reducing contamination by employees.
Packinghouse Equipment
Packinghouse equipment should always be maintained in clean condition. The remnants of product left on belts, tables, lines and conveyors could provide a source for microbial growth; therefore, cleaning by scrubbing to remove particles should be part of the cleaning procedure.
If it is deemed appropriate, sanitizing with a chlorine solution could be accomplished, especially on belt conveyors, and equipment by spot spraying with hand sprayers. Knives, saws, blades, boots, gloves, smocks and aprons should be cleaned or replaced as needed.
Pest Control
A pest control program should be in place to reduce, as much as possible, the risk of contamination by rodents or other animals. In an open or exposed packinghouse operation, the best control is constant vigilance and elimination of any discovered animals and their potential nesting locations. Product and/or product remnants will attract pests; therefore, the daily cleaning of the packinghouse to eliminate the attractive food source should help in reducing pest activity.
Facility Sanitation
Packinghouse facilities have the potential for developing microbial growth on walls, tunnels, ceilings, floors, doors and drains. Scheduled wash down and/ or sanitizing of the facility will reduce the potential for microbial growth. The cooling system should be monitored and cleaned as necessary depending on the type of system.
Temperature Control
Maintenance of proper holding room temperature could affect product quality and could be a factor in reducing microbial growth. Temperature should be monitored in order to ensure maintenance at established product temperature parameters.
Harvesting Carrots
Carrots require three to five months from seeding to harvest when grown as a winter crop. Carrots should be harvested when they reach the appropriate size for the grade and market desired. For U.S. Extra No. 1 and U.S. No. 1, the diameter at the shoulder should be no less than 3/4 of an inch and no more than 1-1/2 inches. For U.S. No. 1 Jumbo, the diameter should be not less than 1 inch nor greater than 2-1/2 inches. The length for U.S. Extra No. 1, U.S. No. 1 and U.S. No. 1 Jumbo should be no less than 5 inches. Smaller or larger carrots can be harvested as U.S. No. 2 or unclassified. The size for U.S. No. 2 carrots is not less than 1 inch and not more than 3 inches in diameter with a minimum length of 3 inches. For processing, carrots are usually a minimum of 1-1/2 inches in diameter at the shoulder. The processor may also have requirements for carrots grown under contract. Care should be taken to harvest carrots at optimum for the particular market because over-mature carrots can develop woody centers particularly in older varieties. Over-mature carrots are also more prone to breakage during harvest. However, delaying harvest by 7-10 days after the appropriate size has been reached will increase the sugar content in mild climatic conditions. Thus, it may be better to delay harvest by one to two weeks depending on the weather.
Carrots can be harvested and sold as bunched carrots, that is with the tops still attached or with short trimmed tops. The size of tops for bunched carrots can vary from under 12 inches to more than 20 inches based on the length classification. Carrots with short trimmed tops will have their tops trimmed to no more than 4 inches.
The condition of the tops is very important particularly for mechanically harvested carrots. Mechanical harvesters pull the carrot from the ground by the tops. Optimum fertility and disease control will help keep tops in condition for harvesting. Heavy rains can impede harvesting not only by limiting field access with equipment but carrot tops can become matted and difficult to pull, resulting in lower yields due to missed carrots.
Carrots grown in Georgia are generally mechanically harvested where the tops are removed by the harvester before being conveyed to a bulk loader. Care should be exercised that the topping mechanism is properly set so that all of the tops are removed. Tops partially remaining can be a site of disease entry as well as transpiring water from the carrot reducing weight and turgor. Processing carrots may be harvested by a potato digger. For this method of harvesting, tops are mowed before digging.
Marketing Carrots
Marketing carrots or any vegetable is more than selling. Marketing includes production, distribution and pricing. To be successful, marketing must be responsive to consumers' demands. Consumers demand quality, freshness, and "reasonable" prices.
Production
Production data for most vegetables are not available or incomplete Most sources of data have California dominating the carrot industry with about 80 percent of the production. About nine other states and imports from Canada and Mexico comprise less than 20 percent of the annual supplies. Carrots are available year-round.
Distribution
The location of the population and their tastes and preferences determine the demand for any product. For most vegetables, the top three markets are the three largest cities -- New York, Los Angeles, and Chicago. This is so with carrots.
Pricing
Supply and demand determine the general price level. The competing states' production determine the supply. Consumers' willingness to buy different quantities at different prices determine the demand.
Consumption data for carrots show increases in all forms -- fresh, canned and frozen. Fresh consumption has recently grown, especially in the baby-peeled pack. An article in The Packer had fresh cut vegetables valued at $690 million in 1996. Growth in fresh cut vegetables grew faster (at 19.5 percent) in 1996 than the growth in salads (16 percent). Fresh-cut carrots are number one at about $480 million. Big growth is expected in baby peeled carrots. (Source: The Packer, A. C. Nielsen scan data). Mann Packing and Grimmway Farms place fresh cut (including carrots) at $1.2-1.3 billion in 1996 (April 7, The Packer). Different sources place baby peeled carrots among the three fastest growing fresh-cut items.
| Table 5. Carrot monthly arrivals and percent of total by state (1996) | ||||||||||||||
| Jan | Feb | Mar | Apr | May | June | July | Aug | Sept | Oct | Nov | Dec | Total | % of Total | |
| 1000 cwt | ||||||||||||||
| California | 497 | 438 | 490 | 509 | 513 | 443 | 455 | 354 | 300 | 370 | 318 | 390 | 5077 | 81.0 |
| Canada | 50 | 49 | 49 | 18 | 4 | - | 1 | 28 | 52 | 56 | 59 | 50 | 416 | 6.6 |
| Florida | 31 | 29 | 39 | 41 | 21 | 10 | -- | - | - | - | - | 5 | 176 | 2.8 |
| Michigan | - | - | - | - | - | - | - | 27 | 45 | 59 | 23 | 3 | 157 | 2.5 |
| Texas | 21 | 14 | 20 | 23 | 19 | 15 | 8 | - | - | - | - | 4 | 124 | 2.0 |
| Washington | - | - | - | - | - | - | 6 | 22 | 24 | 27 | 26 | 14 | 119 | 1.9 |
| Arizona | 6 | 4 | 4 | 11 | 16 | 25 | 14 | - | - | - | - | - | 80 | 1.3 |
| Mexico | - | 2 | 3 | 5 | 14 | 4 | 9 | 12 | 5 | 4 | 7 | 5 | 70 | 1.1 |
| Colorado | - | - | - | - | - | - | 4 | 7 | 11 | 13 | 14 | 3 | 52 | 0.8 |
| Georgia | - | - | - | - | - | - | - | - | - | - | - | - | 0 | 0.0 |
| Total | 605 | 536 | 605 | 607 | 587 | 497 | 497 | 450 | 437 | 529 | - | - | - | 100.0 |
| Calif/total | 82% | 82% | 81% | 84% | 87% | 89% | 92% | 79% | 69% | 70% | 71% | 82% | 81% | - |
| Table 6. Carrots, fresh market: U.S. monthly and season-average grower price, 1990-96 season. | |||||||||||||
| Year | Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec | Annual |
| ---- Dollar per cwt ---- | |||||||||||||
| 1990 | 11.70 | 12.80 | 14.20 | 9.75 | 7.99 | 10.20 | 8.74 | 9.04 | 10.50 | 9.85 | 16.00 | 16.60 | 11.90 |
| 1991 | 21.00 | 13.70 | 16.30 | 13.80 | 13.90 | 11.10 | 9.78 | 10.60 | 10.90 | 11.40 | 19.20 | 17.80 | 14.60 |
| 1992 | 19.00 | 17.10 | 13.20 | 12.80 | 11.70 | 10.80 | 16.90 | 16.70 | 14.40 | 12.80 | 12.00 | 13.80 | 14.50 |
| 1993 | 18.00 | 13.20 | 11.20 | 12.70 | 11.20 | 10.20 | 9.04 | 10.10 | 9.98 | 10.30 | 11.00 | 10.90 | 11.90 |
| 1994 | 10.70 | 10.50 | 11.50 | 10.30 | 12.10 | 12.10 | 13.60 | 16.40 | 15.30 | 15.50 | 15.10 | 15.60 | 12.10 |
| 1995 | 19.20 | 16.90 | 18.90 | 19.80 | 19.30 | 15.30 | 14.80 | 15.50 | 15.90 | 15.10 | 15.10 | 15.40 | 15.30 |
| 1996 | 12.70 | 13.80 | 15.70 | 15.80 | 12.10 | 11.00 | 10.40 | - | - | - | - | - | - |
| Avg | 16.60 | 14.03 | 14.22 | 13.19 | 12.70 | 11.62 | 12.14 | 13.06 | 12.83 | 12.49 | 14.73 | 15.02 | 13.38 |
Grades and Shipping Containers
The are four grades for carrots - U.S. Extra No.1, U.S. No.1, U.S. No.1 Jumbo and U.S. No. 2.
Shipping containers:
50 lb. table carton
48 1-lb. carton
25 lb. table poly bags
24 2-lb. cartons
16 3-lb. cartons
10 5-lb. carton
Baby-peeled
24 1-lb. cartons
20 1-lb. cartons
10 2-lb. cartons
8 5-lb. cartons
73 3-oz carton
Foodservice
50 and 25 lb. poly jumbo
Consumer packs
1, 2, 3, 5, and 10 lb. bags
Value added packs
Cartons of 4 5-lb. bags of shredded carrots, carrot sticks and match sticks (Julienne-cut). Other cuts available are crinkle-cut sticks, diced, sliced, whole peeled or coined.
Summary
Nationally, per capita consumption of carrots is increasing, especially in fresh. This growth in consumption is helping to keep prices from falling.
Production Costs
Enterprise budgets may be used to estimate carrot production costs and break-even prices. The cost estimates included in the budgets should be for inputs necessary to achieve the specified yields over a period of years.
Production practices, size of operation, yields and prices vary among farms, regions and times of the year. For these reasons, each grower should adapt budget estimates to reflect his particular situation. Experience has been shown to be important in achieving high yields. Below, a budget is estimated for carrot production reflecting a reasonably high level of management. (Detailed printed and computerized budgets are available in most county extension offices.)
Type of Costs
Total costs of producing any crop include both variable and fixed costs. The variable or operating costs vary with the amount of crop produced. Common variable costs include seed, fertilizer, chemicals, fuel, and labor. Fixed costs include items such as equipment ownership (depreciation, interest, insurance and taxes), management and general overhead costs. Most of these costs are incurred even if little or no production occurs and are often overlooked for planning purposes.
Variable costs are further broken down into pre-harvest and harvest operations in the budget. This provides the grower an opportunity to analyze the costs at different stages of the production process.
Land cost may either be a variable or a fixed cost. Even if the land is owned, there is a cost involved. Land is included as a fixed cost in this budget. If land is doubled-cropped, each enterprise should be charged half the annual rate. Ownership costs for tractor and equipment (depreciation, interest, taxes, insurance, and shelter) are included as a fixed cost per hour of use. Overhead and management are calculated by taking 15 percent of all pre-harvest variable expenses. This figure is included to compensate for management and farm costs that cannot be allocated to any one specific enterprise. Overhead items include utilities, pick-up trucks, farm shop and equipment, and fees.
Cost Per Unit of Production
The cost categories (Table 7) are broken down in cost per unit at the bottom of the budget. The preharvest variable costs and the fixed costs decline fairly rapidly with increases in yields.
| Costs per hundredweight 1998: | |
| Pre-harvest cost | $0.88 |
| Harvest & marketing cost | $2.75 |
| Fixed cost | $0.32 |
| Total cost | $3.95 |
| (For current cost estimates, see most recent extension vegetable budgets.) | |
Budget Uses
In addition to estimating the total costs and break-even prices for producing carrots, other uses can be made of the budgets.
Estimates of the cash costs (out-of-pocket expenses) provide information on how much money needs to be borrowed. The cash cost estimates are most beneficial in preparing cash flow statements.
In share leases, the cost estimates by item can be used to more accurately determine an equitable share arrangement by the landlord and tenant.
Risk Rated Net Returns
Since there is such a variation in yields and prices from year to year, an attempt is made to estimate the "riskiness" of producing carrots. Five different yields and prices are used in calculating risk. The "expected" values are those prices and yields a particular grower would anticipate to exceed half the time(half the time he would anticipate not to reach these values). Averages can be used for the expected values. "Optimistic" values are those prices and yields a grower would expect to reach or exceed one-year-in-six. The "pessimistic" values are poor prices and yields that would be expected one-year-in-six. The "best" and "worst" values are those extreme levels that would occur "once a lifetime" (1 in 48).
The risk rated section (Table 8) shows there is a 83 percent chance of covering all costs. Half the time, the budgeted grower would expect to net $1280 or more. Half the time he would expect to net less than $1280. One year-out-of-six he would expect: to make more than $2325 per acre or to lose more than $709.
Readers should recognize the examples shown here are estimates. They should serve as guides for developing their own estimates.
| Table 7. Costs and returns expected for carrots, 1998 | |||||
| BEST | OPT | MEDIAN | PESS | WORST | |
| Yield (50# bags) | 1200 | 950 | 700 | 350 | 0 |
| Price per bag | 9.00 | 7.00 | 6.00 | 5.00 | 3.00 |
| Item | Unit | Quantity | Price | Amt/acre | |
| Variable Costs | |||||
| Seed | Lbs | 2.00 | 38.00 | 76.00 | |
| Lime, applied | Ton | 1.00 | 26.00 | 26.00 | |
| Fertilizer | Acre | 1.00 | 133.83 | 133.83 | |
| Calcium/Boron | Gal | 1.25 | 6.50 | 8.13 | |
| Land Plaster | Ton | 0.25 | 35.00 | 8.75 | |
| Fumigant | Gal | 8.30 | 10.00 | 83.00 | |
| Epsom Salts | Acre | 1.00 | 4.50 | 4.50 | |
| Fungicide | Acre | 1.00 | 51.06 | 51.06 | |
| Insecticide | Acre | ||||
| Herbicide | Acre | 1.00 | 49.78 | 49.78 | |
| Other (rye, tissue test, etc.) | Acre | 1.00 | 2.75 | 2.75 | |
| Machinery | Hr | 4.35 | 10.33 | 44.94 | |
| Labor | Hr | 5.50 | 6.00 | 33.00 | |
| Land Rent | Acre | 1.00 | 0.00 | 0.00 | |
| Irrigation | Appl | 6.00 | 5.95 | 35.70 | |
| Interest on Operating Capital | $ | 557.43 | 10.00% | 27.87 | |
| PreHarvest Variable Costs | 585.30 | ||||
| Harvest and Marketing Costs | |||||
| Harvest | Acre | 1 | 29.79 | 29.79 | |
| Haul | Bag | 700 | 0.095 | 66.50 | |
| Grading and Packing | Bag | 700 | 2.00 | 1400.00 | |
| Marketing 8.00% | Bag | 700 | 0.48 | 336.00 | |
| Total Harvest and Marketing | 1832.29 | ||||
| Total Variable Costs | 2417.59 | ||||
| Fixed Costs | |||||
| Machinery and Irrigation | Acre | 1.00 | 86.36 | 86.36 | |
| Land | Acre | 1.00 | 40.00 | 40.00 | |
| Overhead and Management | $ | 585 | 0.15 | 87.80 | |
| Total Fixed Costs | 214.15 | ||||
| Total Budgeted Cost per Acre | 2631.74 | ||||
| Costs per Bag | |||||
| Preharvest variable cost per bag | .88 | ||||
| Harvest and marketing cost per bag | 2.75 | ||||
| Fixed costs per bag | 0.32 | ||||
| Total Budgeted Cost per Bag | 3.95 | ||||
Status and Revision History
In Review on Feb 24, 2009
Published on Apr 01, 2009
Published with Full Review on Apr 25, 2012
