- Foreword
- History, Utilization and Botany
- Culture and Variety Selection
- Soil and Fertilizer Management
- Cabbage Disease Management
- Insect Management
- Weed Management
- Sprayers
- Irrigation
- Harvest and Handling
- Production Costs
- Marketing Cabbage and Leafy Greens
- Acknowledgments
Foreword
This publication is the result of a joint effort among the seven disciplines in the University of Georgia College of Agricultural and Environmental Sciences that serve the Georgia vegetable industry. The 11 topics covered in this bulletin are all integral parts of a successful cabbage/leafy greens management program. Each topic focuses on a particular aspect of production and provides information on the latest management technology for that phase of production. It is hoped that the information contained in this publication will assist growers in improving profitability. Chemical pest control recommendations are subject to change from year to year; thus, only general pest control guidelines are mentioned in this publication. Growers are urged to consult the current Georgia Pest Management Handbook or check with their local county Extension agent regarding the most recent chemical recommendations. Mention of trade names in this publication is neither an endorsement of a particular product nor a lack of endorsement for similar products.
History, Utilization and Botany
Cabbage and leafy greens, including turnip, mustard, kale and collard, have a long history of production in Georgia. The cabbage and leafy greens industry makes up almost 20 percent of the Georgia vegetable industry's acreage, with almost 30,000 acres under production in a given year. The nature of the industry has changed, however, with more emphasis on shipping and less on locally marketed product. The bulk of all these crops remains in the fresh market, with a growing segment of processed greens.
The botanical classifications of several types of cabbage and leafy greens grown in Georgia are listed in Table 1. Brassica oleracea includes all cabbage, collard and kale. This group has long been referred to collectively as "cole crops." This term comes from a Middle English or Norse word that originated from the Latin word caulis, which refers to the cabbage stem or stalk. Wild types of these crops have been found along the Atlantic Coast of Europe and cabbage, kale and collard are believed to have originated in Western Europe. Early uses of these crops were for medicinal purposes.
Table 1. Botanical classification of several types of cabbage and leafy greens
| Common Name | Genus | Species | Group | Subgroup |
| Green Cabbage | Brassica | oleracea | Capitata | Alba |
| Red Cabbage | Rubra | |||
| Savoy Cabbage | Sabauda | |||
| Kale | Acephala | none | ||
| Collard | none | |||
| Mustard | juncea | Pervirdis | none | |
| Turnip | rapa | Rapifera | none |
Kale is thought to be the first form to be domesticated and may have been cultivated as early as 2000 BC. Hard-headed types of cabbage were not cultivated until around the ninth century. Traders and explorers spread the crops to other parts of the world, where they were quickly adopted as cultivated crops. Although cabbage is not particularly high in protein, vitamins and minerals, kale is one of the most nutritious vegetables grown, based on fresh weight. Mustard originated in central and eastern Asia, the Mediterranean and the Himalayas.
Uses of cabbage and leafy greens vary widely. All are grown for both the fresh market and processing into any number of products. Cabbage is popular as a fresh item used in slaw, cooked or processed into preserved products such as sauerkraut. Kale is used primarily as a garnish because of its attractive curly leaves. However, kale can be cooked and eaten in much the same fashion as other leafy greens. Collard, turnip greens and mustard are used primarily as cooked fresh vegetables. However, the tendency to find them served in a raw form is increasing. All are grown for the fresh market but also are grown widely for processing both as canned and frozen products.
Collard, kale, turnip and cabbage are dicotyledonous herbaceous plants that are biennial in nature, although the wild form of cabbage is an annual. All are grown commercially as annuals, however. Kale, turnip and collard do not form a head; all have a rosette form of vegetative growth on a short stem. Collard produces a large smooth leaf, and turnip produces smaller leaves that are generally pubescent. Newer varieties of turnip may be smooth-leaved and are more similar in appearance to mustard. Mustard is a cool-season annual that also forms a rosette of vegetative growth. Mustard leaves may be curly or smooth depending on the cultivar. Cabbage forms a head as the leaves mature and become densely packed.
Cabbage and leafy greens are all produced for their leaves. Some types of turnip also produce a fleshy root harvested for consumption. These roots can vary in shape from flat-round to elongated to spherical. The outer color may be totally white or have shades of red or purple, particularly at the shoulder. Cabbage, collard and kale are hard to distinguish from one another at the seedling stage but soon develop recognizable characteristics.
All of these crops may be subject to bolting (premature appearance of a flower/seed stem). Flowering usually occurs after an extended period of exposure to low temperatures (below 50°F) followed by a period of warmer temperatures. Exposure to temperatures below 40°F or above 70°F after low temperature exposure causes rapid emergence of the seed stem. Losses to bolting in these crops occur almost every year, particularly in overwintered crops that are exposed to severe temperatures. The flowers of the crops are whitish or yellow and are pollinated by insects.
Culture and Variety Selection
Climatic Requirements
Cabbage and leafy greens are adapted to a wide range of environmental conditions and are grown throughout Georgia. Although the southwestern portion of the state produces the most, several areas contribute significantly, including the southeast and the northeast. Cabbage, collard and kale can tolerate hard frosts, but severe freezes can be damaging. Turnip and mustard can tolerate fairly cold temperatures, but hard frosts can kill the crops or make them unmarketable. All can be grown on a wide range of soil types and are somewhat drought tolerant, although production without irrigation is not recommended.
Most production of cabbage and leafy greens occurs in the spring, fall and winter months, except in the northern reaches of the state where production occurs in spring and summer. Collard production in the summer in South Georgia is generally difficult because of increased disease and insect pressure. However, many producers choose to grow it throughout the year. Mustard and turnip can be grown practically throughout the year in Georgia, but cooler temperatures make winter production unlikely in North Georgia. Heat, disease and insect pressure in the summer reduce production in South Georgia.
Crop Establishment
Turnip and mustard are direct seeded. Kale, collard and cabbage can be either direct seeded or transplanted. Transplanting has some advantages over direct seeding but also involves an increased cost. When purchasing transplants, growers should always buy Georgia-certified plants from reputable growers.
Producers of containerized plants specialize in growing plants in greenhouses that are designed specifically for the production of transplants. To contract with a grower for transplants, specify the cell size desired, the variety to be planted and a specific delivery date of the plants.
Also, determine whether the plant grower or the greens grower is to furnish the seed. The cost to the grower for this type of transplant will vary depending on the volume ordered and the cell size of the tray. Growing containerized transplants is a highly skilled, intensive operation that is usually not economically feasible for the greens producer.
Although some greens are seeded directly in the field, several recommended practices should be considered. Direct seeding has several problem areas that must be addressed:
- Weed control is usually somewhat more difficult with direct seeding.
- Direct seeding requires well-made seedbeds and specialized planting equipment adequate to control depth of planting and in-row spacing. Precision seeders that maximize singulation should be considered, although several types of seeders will do an adequate job.
- Because of the shallow planting depth required, the field must be nearly level to prevent seeds from being washed away or covered too deeply with water-transported soil.
- Spring harvest dates will be at least four weeks later for direct-seeded greens. At soil temperatures of 59°, 68° and 77°F, cabbage seeds require 15, nine and six days, respectively, for emergence. Turnip will emerge at soil temperatures of 50°, 59°, 68° and 77°F in five, three, two and one day, respectively, from seeding.
Typically, four- to six-week old cabbage, kale or collard seedlings are transplanted into the field. As with most other vegetable crops, field grown (bare-root) or container-grown transplants may be used. Container-grown transplants retain transplant growing media attached to their roots after they are removed from the container (flat or tray). Many growers prefer this type of transplant because it:
- is less subject to transplant shock,
- usually requires little, if any, replanting,
- resumes growth more quickly after transplanting, and
- grows and produces more uniformly.
Cabbage, collard and kale, like other transplants, should be hardened off before they are transplanted in the field. Hardening off is a technique used to slow plant growth prior to field setting so the plant can more successfully withstand unfavorable conditions in the field.
Cabbage, collard and kale transplants are sensitive to environmental conditions. Any condition that results in a prolonged cessation or checking of vegetative growth during the early stages of plant development can trigger the onset of bolting. Bolting is the development of small, unmarketable heads or flower stalks while the plant is still immature. Flower stalks can form when plants are grown below 50°F in the bed and are exposed to periods of cool weather (35° to 50°F) after field setting. Lack of nitrogen or other nutrient stresses as well as competition from weeds, insects or diseases that slow vegetative growth can promote flowering. Transplants that are older and less vigorous are more likely to flower than young, fast-growing plants. Bare-rooted plants that have been exposed to drying or severe water stress immediately following transplanting are also more likely to flower.
Flowering (heading out) can be prevented by:
- Maintaining a steady, moderate rate of growth in the plant bed.
- Setting out young, healthy transplants that have not been stressed.
- Using transplant water to initiate root growth and remove air pockets from the soil.
- Setting out plants under favorable conditions for growth and irrigating to relieve moisture stress.
- Maintaining good vegetative growth, particularly for the first few weeks after field setting.
Cabbage and collard transplants should never have flower buds at transplanting. An ideal transplant is young (4 inches tall with a stem approximately 1/8 inch in diameter), exhibits rapid vegetative growth, and is slightly hardened at transplanting time. Hardening may be indicated in the greens by a slight purpling of the outer part of the leaves. Good growth following transplanting helps assure a well-established plant.
Transplants should be set out as soon as possible after they are removed from their containers or pulled. If greens transplants must be held for several days before transplanting them, keep them cool (around 55° to 65°F, if possible) and prevent the roots from drying out prior to transplanting. When setting out plants, the transplant should be planted deeply enough to completely cover the rootball (slightly deeper than they were grown).
At transplanting, an appropriate fertilizer starter solution should be applied (see the section on fertilizer starter solutions). After transplanting (especially within the first two weeks), it is very important that soil moisture be maintained so that plant roots can become well established.
Plant Spacing and Planting Dates
The optimal plant population per acre depends upon the plant's growth habit (compact, medium or spreading), size (small, medium or large) at maturity, vigor of specific cultivars, climate, soil moisture and nutrient availability, soil productivity and intended use. Table 2 gives planting dates and recommended seeding and planting information for collard, cabbage, kale, turnip and mustard.
Plant spacing for cabbage and kale is usually about 36 inches between rows and 9 to 12 inches in the row. Turnip may be spaced closely (four to six seeds per foot) if no roots are to be harvested or spread to wider in-row spacings (three to four seeds per foot) if grown for roots and tops. One popular arrangement is to grow four rows, each 14 inches apart, on a 6-foot bed. Mustard arrangements are similar to turnip grown for greens only.
Collard spacing will depend on the harvest method. If young collards are to be harvested similarly to turnip, they may be grown in rows 12 to 18 inches apart with plants 2 to 4 inches apart in the row. If they are to be cropped or cut as whole plants, they should be spaced in rows 36 inches apart with plants 12 to 18 inches apart. Kale can be grown in 30- to 36-inch rows or with three rows on a 6-foot bed with plants 9 to 12 inches apart. Cabbage grown for sauerkraut or slaw may be spaced further apart in the row.
Planting dates can vary widely. However, those crops that are planted between late October and mid-February in South Georgia can come under significant risk of freeze injury. Although successful collard and cabbage production can be achieved with winter plantings, turnip, mustard and kale are more subject to cold damage.
Table 2. Recommended planting dates and planting information for cabbage, collard, kale, turnip and mustard produced in Georgia
|
|
Turnip | Mustard | Collard | Kale | Cabbage |
| Planting Dates | |||||
| North Georgia | March-Aug. | March-Aug. | Feb.-July | March-Aug. | Feb.-July |
| South Georgia | Aug.-Oct. Feb.-April |
Aug.-Oct. Feb.-April |
Aug.-June | Aug.-Oct. Feb.-April |
Aug.-March |
| Planting Information | |||||
| Seed/Pound | 240,000 | 240,000 | 144,000 | 144,000 | 144,000 |
| Seed/Acre, Drilled | 1-2 pounds | 2-3 pounds | 1-2 pounds | 1-2 pounds | 1 pounds |
| Seed/Acre, Precision | 1 pound | 1 pound | ¼ pound | ¼ pound | ¼ pound |
| Seed/Acre, Container Transplants | NA | NA | 2-3 ounces | 2-3 ounces | 2-3 ounces |
| Field Transplants | NA | NA | 6-8 ounces | 6-8 ounces | 6-8 ounces |
| In-row Spacing | ½"-6" | 5"-10" | 2"-18" | 8"-12" | 9"-12" |
| Between-row Spacing | 12"-36" | 12"-36" | 12"-36" | 18"-36" | 30"-42" |
| Seeding Depth | 1/8"-¼" | 1/8"-¼" | ¼" | ¼" | 1/8"-¼" |
| Seed to Harvest | 35-50 days | 35-50 days | 60-90 days | 60-80 days | 90-140 days |
| Transplant to Harvest | NA | NA | 6-10 weeks | 6-8 weeks | 70-120 days |
| Optimum Soil Temperature for Germination (°F) | 60-85 | 60-85 | 50-85 | 50-85 | 50-85 |
| Optimum Air Temperature for Crop Growth (°F) | 60-65 | 60-65 | 60-65 | 60-65 | 60-65 |
Variety Selection
Numerous varieties of cabbage and leafy greens are available on the market. Several factors should be considered in selecting appropriate varieties. Yield, of course, is important to every grower. However, this should not be the sole factor in determining variety. Disease resistance/tolerance is important in many of these crops and should be critically examined. Buyer preference and market acceptability are probably two of the most important factors to consider. Horticultural characteristics such as product color, growth habit and shape should also be considered. Finally, the variety should be adapted to the area in which it is to be grown.
Local variety trials are a good source of information regarding variety selection. With any new variety, always try a small planting of the variety first before adapting it to your operation. Also, give every new variety at least a couple of tries before making a decision on its use for your production system. Environmental conditions can strongly influence varietal performance. Therefore, conditions in one year may not produce the same results in another year for a given variety. Good varieties are adaptable over a wide range of conditions. Tables 3 and 4 show some recommended varieties for Georgia production of cabbage and leafy greens.
Table 3. Cabbage varieties recommended for production in Georgia
| Variety | Color | Head Size | Maturity | Shape |
| A&C No. 5 Plus | blue-green | medium | 91 days | flat-round |
| Blue Bayou | blue-green | medium | 78 days | semi-round |
| Blue Vantage | blue-green | 4-6 pounds | 72 days | round |
| Bravo | blue-green | 4-8 pounds | 85 days | round |
| Constanza | blue-green | 4-6 pounds | 90 days | round |
| Crimson | red | small-medium | 82 days | round |
| Fortuna | green-blue | 4-6 pounds | 90 days | round |
| Hercules | blue-green | medium | 91 days | flat-round |
| Izalco | blue-green | medium | 105 days | round |
| Quisto | blue-green | medium | 89 days | round |
| Red Dynasty | red | medium-large | 70 days | round-oval |
| Red Rookie | red | 3 pounds | 78 days | round |
| Rio Verde | blue-green | 85 days | flattened | |
| Royal Vantage | blue-green | 3 pounds | 102 days | round |
| Savoy Ace | dark green | 2 pounds | 80 days | round |
| Savoy King | green | 4 pounds | 111 days | semi-flat |
| Solid Blue No. 780 | blue-green | 3-4 pounds | 78 days | round |
| Solid Blue No. 790 | blue-green | 3-4 pounds | 79 days | round |
Table 4. Varieties of collard, turnip, mustard and kale recommended for production in Georgia
|
Collard
|
Turnip
|
Mustard
|
Kale
|
|
Blue Max
Champion Flash Georgia Heavi Crop Top Bunch Vates |
Alamo
All Top Just Right Purple Top Seven Top Shogoin White Globe |
Florida Broadleaf
Slobolt Southern Giant Curled Tendergreen
|
Blue Armor
Blue Knight Vates |
Soil and Fertilizer Management
Georgia's climate, stretching from the warm Coastal Plain in the south to the cool mountain area in the northern region of the state, provides a long growing season for producing cabbage and leafy greens. Although these crops grow best in light, fertile, well-drained soils, when production is properly managed, they can be grown successfully in a wide range of soil types throughout Georgia.
Soil Management
Plants depend on the soil for physical support, nutrients and water. The degree to which the soil adequately provides these factors depends upon topography, soil type, soil structure and soil fertility. Under cultivated conditions, soil and fertilizer management are two key factors influencing plant growth and yield.
Tillage is a general term for any operation that disrupts and/or moves the soil, typically within 10 to 12 inches of the soil surface. Land preparation involves one or more tillage operations that loosens, pulverizes, smooths or firms the soil and makes it more conducive to plant establishment and root growth.
Growth of cabbage and leafy green roots is influenced (and in many cases is limited) by the soil profile. Hard pans, clay pans and generally compacted soil restrict root growth. This, in turn, reduces nutrient and water uptake, limits plant growth and reduces yields. Although cabbage and leafy greens are shallow rooted, under favorable conditions and in properly prepared soil, roots will grow to a depth of 18 to 24 inches.
Tillage with a moldboard ("bottom") plow provides the greatest soil volume conducive to vigorous root growth. Disking after moldboard plowing recompacts soil.
Compaction pans are present in many Georgia soils. They are usually formed by machinery and, when present, are normally at or just below plow depths. Even though compaction pans may be only a few inches thick, their inhibitory effects on root growth can significantly reduce yields of greens and cabbage.
If a compaction pan exists just below or near moldboard plow depth, disrupting this hard pan by subsoiling to a depth of 16 to 18 inches will allow the development of a more extensive root system and increase water infiltration.
If a lot of plants or plant residue is on the soil surface, disking (or mowing, then disking) is helpful before moldboard plowing. Immediately prior to seeding or transplanting, final soil preparation (to ensure a crust-free, weed-free friable soil) can be accomplished with a rotary tiller, bed press, bedding disk or double disk hiller with a leveling board.
Cabbage and leafy greens may be planted or transplanted on flat or raised beds. A raised bed will warm up more quickly and enhance earlier growth. Cabbage and leafy greens do poorly in excessively wet soils. Raised beds facilitate drainage and help prevent "wet feet" in low or poorly drained soils. Keep in mind, however, that cabbage or leafy greens planted on raised beds may require more irrigation during drought conditions.
Lime and fertilizer management refers to the application of optimal amounts of lime and fertilizer (or nutrient-containing materials) at the most appropriate time(s) and by the most effective method.
Fertilizer Management
Recommendations based on soil tests result in the most effective lime and fertilizer management program possible. However, recommendations can be accurate only if valid soil sampling procedures are used to collect the samples submitted for analysis. To be beneficial, a soil sample must reliably represent the field or "management unit" from which it was taken. Improperly collected, compiled or labeled soil samples are of dubious benefit and may even be detrimental. If you have questions about soil sampling, please contact your county Extension office for more information. Recent technological advances have made it possible to use quadrant sampling or global positioning techniques to tune fertility recommendations more finely, which can result in more uniform fertility throughout a given field.
Recommending a specific fertilizer management program that has universal application for all cabbage and leafy greens fields is impossible. In addition to crop nutrient requirements and soil types, fertilizer recommendations should take into consideration soil pH, residual nutrients and inherent soil fertility. Therefore, fertilizer recommendations based on soil analysis have the greatest potential for providing cabbage and leafy greens with adequate, but not excessive, fertility. Apply only the needed amounts of fertilizer for optimum growth and yield. Excessive fertilizer application wastes fertilizer, encourages luxury consumption of plant nutrients and may cause fertilizer burn
Soil pH
Soil pH influences plant growth, the availability of nutrients and the activities of microorganisms in the soil. Keeping soil pH in the proper range is important for production of the best yields of high quality cabbage and leafy greens. Soil test results indicate soil pH levels and are used to recommend the amount of lime required to raise the pH to the desired range.
The optimum pH range for cabbage and leafy greens production is 6.0 to 6.5. Coastal Plain soils, which predominate in South Georgia, become strongly acid (pH 5 or less) with time if lime is not applied. A soil test is essential for determining how much lime should be applied.
Calcium (Ca) has limited mobility in soil; therefore, lime should be broadcast and thoroughly incorporated to a depth of 6 to 8 inches to neutralize the soil acidity in the root zone. To allow adequate time for neutralization of soil acidity (raising the pH), lime should be applied and thoroughly incorporated two to three months before seeding or transplanting. However, if application cannot be made this early, liming will still be beneficial if applied and incorporated at least one month prior to seeding or transplanting.
Generally, maintaining a soil pH of 6.0 to 6.5 will provide adequate soil test Ca levels. When soil test Ca levels fall below 350 to 400 pounds per acre, calcium deficiency problems may develop. Because calcium levels can vary considerably in a field, soil test calcium levels on Coastal Plain soils should be maintained at 500 pounds per acre or slightly above. If the soil pH is between 6.0 and 6.5 and the soil test Ca level drops below 500 pounds per acre on Coastal Plain soils, apply 800 to 1,000 pounds of calcium sulfate (gypsum) per acre.
Two liming materials commonly available in Georgia are calcitic and dolomitic limestone. In addition to calcium, dolomitic limestone contains 6 percent to 12 percent magnesium. Because Coastal Plain soils are inherently low in magnesium, dolomitic limestone is usually the preferred liming material.
Phosphorus and Potassium Recommendations
The following chart indicates the pounds of fertilizer nutrients recommended for varying soil fertility levels according to University of Georgia soil test ratings of residual phosphorus (P) and potassium (K).
Fertilizer Recommendations for Cabbage and Leafy Greens
| Ratings | Low | Medium | High | Very High |
| Recommended P | 160 | 110 | 60 | 0 |
| Recommended K | 160 | 110 | 60 | 0 |
P – pounds of P2O5 recommended per acre
K – pounds of K2O recommended per acre
NOTE: If soil testing is done by a lab other than the University of Georgia Soil Testing Laboratory, the levels recommended above may not apply.
All the recommended phosphorus should be applied during or near the time of transplanting. One-third to one-half of the potassium should either (1) be applied in two bands, each 2 to 3 inches to the side and 2 to 3 inches below the level of plant roots or (2) be incorporated into the bed prior to seeding or transplanting. Broadcasting over the entire field is usually less effective than banding. An acceptable alternative to field broadcasting is the "modified broadcast" method by which a preplant fertilizer containing one-third to one-half of the nitrogen and potassium and all the recommended phosphorus and micronutrients is broadcast in the bed area only. For example, on a 72-inch-wide bed, a swath (60 to 72 inches wide) of fertilizer is uniformly applied centered over the bed. Incorporation by rotary tilling will help reduce water and wind movement of the fertilizer and will also place some fertilizer in the root zone. The remainder of the recommended nitrogen and potassium should be applied in one to three applications as needed. It can be banded in an area on both sides of the row just ahead of the developing root tips. On sandy soils, the maximum number of applications is usually more effective.
Nitrogen Recommendations
For production of cabbage and leafy greens, Coastal Plain soils require 175 to 225 pounds of nitrogen (N) per acre. Extremely sandy soils may need additional N or an increased number of applications. Increasing the number of applications may prove beneficial because it will cut down on the amount lost to leaching from adding too much fertilizer at one time. Piedmont, Mountain and Limestone Valley soils usually require 150 to 180 pounds of N per acre for cabbage and greens production.
Required N rates will vary depending on season rainfall, soil type, soil temperature, irrigation, plant population, and method and timing of applications.
For typical Coastal Plain soils, one-fourth to one-third of the recommended nitrogen should be incorporated in the bed prior to seeding or transplanting. Broadcasting over the entire field is usually less effective than banding.
An acceptable alternative to field broadcasting is the "modified broadcast" method (described under Phosphorus and Potassium Recommendations). Incorporation by rotary tilling will help reduce water and wind movement of the fertilizer and will place some fertilizer in the root zone. Apply the remaining recommended N in one to three applications as needed. It can be banded in an area on both sides of the row just ahead of the developing root tips. For heavier Piedmont, Mountain and Limestone Valley soils, one or two applications are usually sufficient.
Magnesium, Sulfur, Zinc and Boron Recommendations
If the soil test indicates magnesium is low and if lime is recommended, apply dolomitic limestone. If magnesium is low and lime is not recommended, apply 25 pounds of elemental magnesium per acre. Apply a minimum of 10 pounds of sulfur per acre, 1 pound of actual boron per acre, and, if soil test indicates zinc is low, 5 pounds of actual zinc per acre. Sulfate of potash magnesia (Sul-Po-Mag or K-Mag) may be used to supply a portion of the recommended K20 and to supply magnesium (Mg) and sulfur (S).
Starter Fertilizer Solution
Liquid fertilizers applied to the soil around plant roots are called starter solutions. They promote rapid root development and early plant growth. Starter solutions for cabbage and leafy greens transplants should contain a high rate of phosphorus (an approximate ratio of 1 nitrogen: 3 phosphorus: 0 potassium is common) and should be mixed and applied according to the manufacturer's directions. Most starter solutions consist of 3 pounds of a formulated material (such as 10-34-0, which weighs approximately 11 pounds/gallon) mixed in 50 gallons of water. Approximately ½ pint of the starter solution is normally applied per plant. Application of about 100 to 150 pounds per acre of a starter fertilizer promotes earlier growth, particularly in cool/cold soils. For early growth stimulation, pop-up fertilizer should be banded 2 to 3 inches to the side of the plants and 2 to 3 inches below the roots. In addition to supplying phosphorus, which has limited availability in cold soils, the starter solution supplies water and firms the soil around roots (eliminating air pockets, which cause root drying). However, a starter solution is no substitute for adequate rainfall or irrigation after transplanting.
Mix and apply starter fertilizer according to the manufacturer's recommendations. If it is too concentrated, a starter solution can kill plant roots and result in dead or stunted plants. When mixing and applying from a large tank, mix a fresh solution only after all the previous solution is used from the tank. This helps prevent the gradual increase in concentration that occurs when a portion of the previous mix is used for a portion of the water component in subsequent batches.
Foliar Fertilizer Application
The fact that plants can absorb a number of fertilizer elements through their leaves has been known for some time. However, leaves of many vegetable plants are not especially well adapted for absorbing nutrients because they have a waxy cuticle. In fact, plants may appear to benefit from foliar uptake when the actual cause of improvement may be from the component of the nutrient spray that reaches the soil and provides essential nutrients for subsequent root uptake.
The effectiveness of applying macronutrients such as nitrogen, phosphorus and potassium to plant leaves is questionable. It is virtually impossible for greens (waxy leaved cabbage, collard and kale) to absorb enough N, P or K through their leaves to meet their nutritional requirements; furthermore, they are unlikely to absorb sufficient amounts of macronutrients to correct major deficiencies. Although nitrogen may be absorbed within 24 hours after application, up to four days are required for potassium uptake, and seven to 15 days are required for phosphorus to be absorbed from foliar application.
The crucial question is whether foliar N, P or K actually increases yield or enhances quality. Although some growers feel that foliar fertilizer should be used to supplement a soil-applied fertilizer program, research findings do not support this practice. If proper fertilizer management of soil-applied nutrients is used, supplementation by foliar fertilization is not usually required.
Foliar nutrients often are expected to cure a variety of plant problems, many of which may be unrelated to nutrition, such as reducing stress, aiding in healing frost- or hail-damaged plants or increasing plant resistance to various stresses and pests. Nutrients are effective as long as they are supplying a nutritional need; however, neither soil-applied nor foliar-applied nutrients are capable of performing miracles.
After frost or hail occurs, some cabbage and leafy greens growers apply foliar nutrients to give the plants an "extra shot" to promote rapid recovery. However, if a proper fertilizer program is being used before foliage damage occurs, the plants don't need additional fertilizer. What they do need is time and the proper environment for the normal recovery processes to occur. The likelihood of deriving significant nutritional benefits from a foliar application of fertilizer to plants that have lost some of their leaves (or have a large proportion of their leaves severely damaged) is questionable.
Foliar application of sulfur, magnesium, calcium and micronutrients is helpful in correcting deficiencies. To alleviate boron deficiency, apply water-soluble boron (l lb/boron per acre). Magnesium sulfate (3 lb/acre) can be applied to correct a magnesium deficiency. CaB, a commercially available formulation containing 2 percent boron and 6 percent calcium, can be applied at the rate of 2 quarts per acre to help alleviate calcium and boron deficiencies. Other acceptable foliar fertilizer formulations may also be available. When using foliar fertilizers (1) check the label for percent active ingredients - know what and how much fertilizer you are applying - and (2) apply according to label instructions. Apply foliar fertilizers only if a real need for them exists and only in quantities recommended for foliar application. Application of excessive amounts can cause fertilizer burn and/or toxicity problems.
Tissue Analysis
Fertilizer recommendations provide general guidelines for the application of fertilizers to crops. Actual amounts needed will vary depending on soil type, the amount of leaching and crop growth. Routine tissue analyses (tables 5 and 6) and sap tests (Table 7) are excellent tools for fine tuning fertilizer management.
Presently we do not have tissue and sap tests recommendations for all vegetable crops. We will provide recommendations for other vegetable crops when they are developed.
Table 5. Plant tissue analysis critical values for cabbage
| Status | Nutrient | |||||||||||
| N (%) | P (%) | K (%) | Ca (%) | Mg (%) | S (%) | Fe (ppm) | Mn (ppm) | Zn (ppm) | B (ppm) | Cu (ppm) | Mo (ppm) | |
| Five Weeks After Transplanting1 | ||||||||||||
| Deficient | <3.2 |
<0.3 |
<2.8 |
<0.8 |
<0.25 |
– |
<30 |
<20 |
<30 |
<20 |
<3.0 |
<0.3 |
| Adequate Range |
3.2-6.0 |
0.3-0.6 |
2.8-5.0 |
1.1-2.0 |
0.25-0.6 |
0.3-0.6 |
30-60 |
20-40 |
30-50 |
20-40 |
3.0-7.0 |
0.3- 0.6 |
| High | >6.0 |
>0.6 |
>5.0 |
>2.0 |
>0.6 |
– |
>100 |
>40 |
>50 |
>40 |
>10 |
– |
| Eight Weeks After Transplanting1 | ||||||||||||
|
Deficient |
<3.0 |
<0.3 |
<2.0 |
<0.8 |
<0.2 |
0.3 |
<30 |
<20 |
<30 |
<20 |
<3.0 |
<0.3 |
|
Adequate |
3.0-6.0 |
0.3-0.6 |
2.0-4.0 |
1.0-2.0 |
0.25-0.6 |
0.3-0.8 |
30-60 |
20-40 |
30-50 |
20-40 |
3.0-7.0 |
0.3-0.6 |
|
High |
>6.0 |
>0.6 |
>4.0 |
>2.0 |
>0.6 |
0.8 |
>100 |
>40 |
>50 |
>40 |
>10 |
0.6 |
| Heads One-half Grown2 | ||||||||||||
| Deficient | <3.0 | <0.3 | <1.7 | <0.8 | <0.25 | – | <20 | <20 | <20 | <30 | <4.0 | <0.3 |
| Adequate Range |
3.0-4.0 | 0.3-0.5 | 2.3- 4.0 | 1.5- 2.0 | 0.25-0.45 | 0.3 | 20-40 | 20-40 | 20-30 | 30-50 | 4.0-8.0 | 0.3-0.6 |
| High | >4.0 | >0.5 | >4.0 | >2.0 | >0.45 | – | >100 | >40 | >40 | >50 | >10 | – |
1 Most recently mature leaf sampled.
2 Wrapper leaf sampled.
Adapted from Plant Tissue Analysis and Interpretation for Vegetable Crops in Florida. University of Florida Publication SS-Vec-42. January 1991.
Table 6. Injection schedule for N and K for cabbage and collard planted two rows per 6-foot bed on soils testing medium for K
| Crop |
Total Nutrients (lb/A) |
|
Crop Development | Injection (lb/A/day)1 | |||
| N | K2O | Stage | Weeks2 | N | K2O | ||
| Cabbage | 175 | 110 | 1 | 3 | 1.5 | 1.0 | |
| Collard | 175 | 110 | 2 | 7 | 2.5 | 1.5 | |
| 3 | 2 | 1.5 | 1.0 | ||||
1 Actual amounts may vary depending on preplant fertilizer and soil test K.
2 Starting from seeding or transplanting. First two weeks may be omitted if 25 percent of total N and K were applied preplant.
Adapted from Vegetable Production Guide for Florida. Pub. No. SP 170. University of Florida Cooperative Extension Service. April 1999.
Table 7. Plant tissue analysis critical values for collard
| Status | Nutrient | |||||||||||
| N (%) | P (%) | K (%) | Ca (%) | Mg (%) | S (%) | Fe (ppm) | Mn (ppm) | Zn (ppm) | B (ppm) | Cu (ppm) | Mo (ppm) | |
| Young Plants1 | ||||||||||||
| Deficient | <4.0 | <0.3 | <3.0 | <1.0 | <0.4 | – | <40 | <40 | <25 | <25 | <5 | – |
| Adequate Range |
4.0-5.0 | 0.3-0.6 | 3.0-5.0 | 1.0-2.0 | 0.4-1.0 | – | 40-100 | 40-100 | 25-50 | 25-50 | 5-10 | – |
| High | >5.0 | >0.6 | >5.0 | >2.0 | >1.0 | – | >100 | >100 | >50 | >50 | >10 | – |
| At Harvest1 | ||||||||||||
|
Deficient |
<3.0 | <0.25 | <2.5 | <1.0 | <0.35 | <0.3 | <40 | <40 | <20 | <25 | <5 | <0.3 |
|
Adequate |
3.0- 5.0 | 0.25-0.5 | 2.5-4.0 | 1.0-2.0 | 0.35-1.0 | 0.3-0.8 | 40-100 | 40-100 | 20-40 | 25-50 | 5-10 | 0.3-0.8 |
|
High |
>5.0 | >0.5 | >4.0 | >2.0 | >1.0 | >0.8 | >100 | >100 | >40 | >50 | >10 | >0.8 |
1 Tops of young plants sampled.
2 Most recent mature leaf sampled.
Adapted from Plant Tissue Analysis and Interpretation for Vegetable Crops in Florida. University of Florida Publication SS-Vec-42. January 1991.
Table 8. Sufficiency ranges for petiole sap testing in collard
| Crop Development Stage | Fresh Petiole Sap Concentration (PPM) | |
| NO3-N | K | |
| Six-leaf Stage | 800-1,000 | NR |
| One Week Prior to First Harvest | 500-800 | NR |
| First Harvest | 300-500 | NR |
NR - No recommended ranges have been established.
Adapted from Vegetable Production Guide for Florida. Publication No. SP 170. University of Florida Cooperative Extension Service. July 1996.
Cabbage Disease Management
Cabbage and leafy greens are susceptible to a number of diseases that may seriously injure or even destroy the crop. Some diseases may cause only minor spotting, but because the leaves are consumed, the quality may be reduced below market standards. Prevention is the key to controlling all diseases affecting crucifers. Some of the diseases can be controlled with timely fungicide applications and others must be prevented altogether. This section will aid in the identification of diseases and discuss environmental conditions that favor disease development. Sources of infection are discussed relative to specific diseases on specific crops.
Black-rot(Figure 1) Black-rot, caused by the bacterium Xanthomonas campestris pv. campestris, is the most serious disease of crucifers in Georgia. The bacterium attacks many species of the mustard family. Among these are cabbage, collard, kale, mustard and turnip. Plants may be affected at any stage of growth. This disease is seedborne and is often introduced by contaminated seeds or infected transplants. In some areas of the country, the disease is of minor importance; however, under Georgia conditions, the disease becomes serious and many growers sustain severe economic loss. In some cases, the crop may be destroyed. In the field, the disease is easily recognized by the presence of large yellow to yellow-orange V-shaped lesions extending inward from the margin of the leaf. When infected seeds germinate, the resulting young plants usually die quickly; however, these plants serve as an inoculum source for other plants. If infection occurs in young seedlings, the disease is much more severe because the main stem becomes infected and the disease becomes systemic and moves throughout the plant. These plants remain stunted and the veins in the stem are black. Heads developed from these plants deteriorate rapidly after harvest. The bacterium enters the vascular system of the plant principally through natural openings and injuries on the leaf. In time, the bacterium spreads in the vascular system of the leaf and stem. The disease spreads and causes most damage in wet, warm weather. It does not usually spread in dry weather and is inactivated at temperatures below 50°F. The bacterium can survive in the soil for 14 to 42 days, depending on the season, and in old cabbage stems for 244 days. The disease is also carried over on weed hosts such as "pepper grass" and with wild species of mustard, radish and turnip. The bacterium is spread by splashing rain, irrigation and running surface water as well as insects and other movement in the field while the plants are wet. Disease management strategies such as crop rotation away from contaminated land for two years and use of certified seed or transplants are effective. There is no remedial control for this disease |
 Figure 1. Black-rot. Figure 1. Black-rot. |
Wirestem, Bottom Rot and Head Rot(Figure 2) Wirestem, bottom rot and head rot are caused by Rhizoctonia solani. This fungus also is a common cause of damping-off. Wirestem is normally more serious in transplant beds; however, it can be a serious problem after plants are transplanted to the field. Cabbage, collard and kale planted in early fall are more vulnerable to Rhizoctonia injury than spring plantings. Wirestem infected plants are first infected near the soil surface. The initial infection site may be as small as a pinpoint or extend an inch up the stems. This area shrivels and becomes reddish brown; outer tissues slough off, leaving the woody inner stem exposed. The infected plant may be bent or twisted without breaking — hence the name. The wirestem fungus is common to most Georgia soils. The amount of Rhizoctonia present is greatly influenced by the cropping history. Wirestem damage may be suppressed by adding a fungicide to the transplant water. Frequent irrigations during hot dry periods will reduce the incidence of the disease; water evaporation cools the soil surface. Rotating with grain crops, deep turning and using a fungicide in the transplant water will greatly reduce the incidence of disease. |
 Figure 2. Wirestem, bottom rot and head rot. Figure 2. Wirestem, bottom rot and head rot. |
Black Leg
Black leg, caused by the fungus Phoma lingam, is another disease that can cause serious loss to cabbage. Plants are usually infected in seedbeds. Usually the first symptom is an oval, depressed, light brown canker near the base of the stem. The canker enlarges until the stem is girdled. Circular light brown spots also appear on the leaves. Soon after cankers or spots begin to form, they are marked with numerous black dots that are the fruiting bodies of the fungus. The fungus lives for at least three years in the soil and is carried on and in the seeds. Black leg is influenced by environmental conditions, with the severity of the disease in direct proportion to the amount of rainfall received. The fungus grows well at all temperatures suitable to cabbage.
Alternaria Leafspot(Figure 3) Alternaria leafspot, caused by the fungus Alternaria brassicae, may cause severe damage if left uncontrolled. The first symptom is a small dark spot on the leaf surface. As the spot enlarges, concentric rings, which are common to this disease, develop. Blight spots enlarge progressively and can defoliate a plant if left uncontrolled. Alternaria leafspot is best controlled by applying a fungicide on a schedule throughout the entire growing season. |
 Figure 3. Alternaria leafspot. Figure 3. Alternaria leafspot. |
Downy Mildew of Cabbage, Collard and Kale(Figure 4) Downy mildew, caused by the fungus Peronospora parasitica, can be transferred from the transplant bed; however, it also can be introduced to new field plantings by windblown spores. Plants can be infected at any time during their growing period. Young plants infected early show a white mildew effect mostly on the underside of the leaf; later, a slight yellowing can be observed on the corresponding upper surface. The young leaf, when yellow, may drop off. Older leaves enlarge as they turn tan in color and papery in texture. The fungus produces a mass of gray growth, conidia, on the undersurface of the leaves. The conidia, or seed bodies, are able to float long distances in cool moist air. With favorable weather, they may germinate in three to four hours and produce a new crop of seed bodies on a susceptible host in as few as four days. Moisture and temperature are very important to the reproduction of this disease. Drizzling rains and cool weather are very favorable for disease development. The fungus grows best and disease develops most rapidly when the night temperatures are about 46°F for four or more consecutive nights and the day temperature does not rise much above 75°F. A preventive spray schedule will help control downy mildew. Downy mildew can infect turnip and mustard at any stage of growth. A grayish mold forms in spots and on the undersurface of the leaf. Later, a slight yellowing shows on the upper surface. Infected spots enlarge as they turn tan in color and papery in texture. When the disease is severe, the entire leaf dies. Heavily infected leaves may have a watery appearance, wilt and die before the mold growth is evident. Occasionally, affected leaves will show hundreds of very small, darkened specks. Downy mildew overwinters in turnip roots or on old tops left in the field. The fungi form thick-walled resting spores in the turnip stem. These tiny bodies become mixed with seed at harvest and can be shipped to new locations. Downy mildew can destroy a field of greens within three to four days after it is first noticed. Usually, damage is most severe on cabbage and leafy greens grown in the spring because conditions favoring its development are more likely to occur at this time. |
 Figure 4. Downy mildew. Figure 4. Downy mildew. |
Sclerotinia sclerotiorium(Figure 5) Sclerotinia sclerotiorium is becoming more prevalent in cabbage fields as well as transplant beds and greenhouses. Recently this disease has also been identified on turnip, mustard and kale. The disease usually begins on the lower stem, causing a watery soft rot followed by white, cottonlike growth. Black sclerotia, overwintering structures that give rise to the spore-producing apothecia, develop later. The disease is favored by cool, wet weather, which may result in a massive steamlike cloud of spores being spread throughout an area. The disease can infect cabbage from the seedling stage to maturity. On mature cabbage, the entire head is often covered with the white mycelium and black sclerotia, sometimes referred to as "raisin head." The entire heads melt down, basically leaving a pile of sclerotia. The remarkable thing about this disease is that it does not cause any odor during the destructive process unless other saprophytic organisms invade, which do cause odor. The best defense against this disease is to transplant disease-free transplants and cultivate to cover any sclerotia. No chemical fungicides are recommended. Good rotation practices are usually all that is needed to manage the disease. |
 Figure 5. Sclerotinia sclerotiorium. Figure 5. Sclerotinia sclerotiorium. |
Cabbage Yellows(Figure 6) Cabbage yellows is caused by Fusarium oxysporum f.sp. conglutinans. The cabbage strain severely attacks many varieties of kale, cabbage and collard, and moderately affects turnip. The disease is often confused with black rot because of the similar symptoms. Both diseases cause leaf drop, curving stalks and the formation of buds on leafless stems. Yellows is more likely to produce a curve in the midrib or cause the leaf to grow on only one side. Fusarium can live for several years in the soil without being associated with any plant parts. After the pathogen becomes established, it spreads by rain and by equipment moving from one field to another. The fungus enters root hairs, and spores are produced inside and outside of affected stems. Most currently recommended varieties are resistant to yellows. |
 Figure 6. Cabbage yellows. Figure 6. Cabbage yellows. |
Leafspots of Mustard and TurnipLeafspot of mustard and turnip is most often caused by downy mildew (Peronospora parasitica), white spot (Cercosporella brassicae), anthracnose (Colletotrichem higginisianum) and cercospora leafspot (Cercospora brassicicola). Cercospora Leafspot (Figure 7, left) Cercospora leafspot is also a major problem on mustard and turnip. This disease is sometimes called frog-eye leafspot. Both Cercospora and Cercosporella develop under similar environmental conditions. The disease is most prevalent in a temperature range of 55° to 65°F, with plenty of moisture. Cercosporella Leafspot (Figure 7, center) This fungus is primarily a problem on turnip and mustard. The spots caused by Cercosporella are white and much larger than those caused by Cercospora, and are referred to as pale-spot. White spot causes pale green to grayish white circular to angular spots on leaves. Each spot has a yellowish to brown halo or border around it. Numerous spots may occur on one leaf and merge, killing the entire leaf. White spot overwinters on such plants as turnip, mustard, collard, cabbage and kale. The fungus produces numerous spores in each infected spot on the leaf. The spores are blown long distances by the wind. White spot reproduces and spreads at a temperature of 71° to 86°F. This disease is usually more damaging to the fall greens crop. Anthracnose (Figure 7, right) Anthracnose is often a serious problem on turnip and mustard and can infect kale and collard. The leafspots are small, pale gray desiccated circular spots. The same fungus infects turnip roots when spores fall from the leaves to the roots. The infected areas begin as small sunken dry spots. Under moist conditions, bacterial soft rot develops, destroying the entire root. The fungus overwinters on crop debris and in volunteer plants. The disease is most severe at temperatures of 79° to 86°F, with plenty of moisture. The disease is prevented by rotation, deep turning and fungicide sprays. |
 Figure 7. Cercospora leafspot (left), Cercosporella leafspot (center) and anthracnose. Figure 7. Cercospora leafspot (left), Cercosporella leafspot (center) and anthracnose. |
White Rust(Figure 8) White rust is most often seen on spinach and is caused by Albugo sp. White rust affects other crucifers. It has been observed on mustard in a few instances. The disease overwinters by means of oospores. In perennial hosts, such as horseradish, it persists in crowns and roots. Secondary spread is by conidia (spores), which are carried by air currents. Moisture on the host surface is essential for germination. White rust is recognized by a white growth, usually on the underside of the leaves. White rust is controlled by soil and foliar applied fungicides. |
 Figure 8. White rust of spinach. Figure 8. White rust of spinach. |
Root-knot Nematode(Figure 9) Root-knot nematode is the only nematode of economic importance that affects crucifers. All species of root-knot are considered pests of crucifers. Cabbage, turnip, mustard and spinach are the main crops affected. Stunted growth and chlorosis are the above-ground symptoms. Classic galling of the root system is key for diagnosing root-knot nematode damage. Rotation and chemical treatment are the control practices. |
 Figure 9. Root-knot nematode. Figure 9. Root-knot nematode. |
Viruses(figures 10, 11) Turnip mosaic virus disease is caused by any of many strains of turnip mosaic. These strains infect almost every crucifer, including weeds such as shepherd's purse and charlock. Infected cabbage plants have mottled, distorted leaves. The production of leaf bloom is reduced, and the whole plant is stunted, especially when infected early in the season. Some strains show a darkened ring effect, especially on the older foliage, and irregular dead areas between the veins. This virus may be responsible for a stippling symptom on the outer and inner leaves of cabbage heads. The virus overwinters in perennial weeds. It is carried by many species of aphids. Cauliflower mosaic virus disease is found more frequently on turnip. Plants infected are rarely stunted, but most express a mosaic pattern. The most distinctive symptom is vein clearing. On cabbage, a black stippling symptom develops on the outer leaves of the mature heads. Cabbage mosaic causes black specking of cabbage heads at harvest or during storage. This mosaic is caused by a single or dual infection of viruses. A strain of turnip or cauliflower mosaic virus may be responsible. Infections late in the season cause minor losses, and early infections cause serious losses. The turnip and cauliflower mosaic viruses are transmitted principally by cabbage aphids and green peach aphids. Neither virus is seedborne. Tomato spotted wilt virus (TSWV) can infect cabbage at any stage of growth. The virus is transmitted by thrips and has been identified in Georgia since 1989. |
 Figure 10. Viruses. Figure 10. Viruses. Figure 11. Viruses. Figure 11. Viruses. |
Insect Management
On-farm Components of Insect Management
Researchers, farmers and agricultural consultants often overlook the basic principles of on-farm insect management when faced with insecticide-resistant pests that attack cabbage and leafy greens. Although theoretical principles may not always apply when working on the farm, certain fundamental practices can greatly enhance the chances of effectively controlling these insects.
For example, diamondback moth caterpillar (DBM) resistance to insecticides has been documented for all major insecticide classes: carbamates, organophosphates and pyrethroids. Although very few currently registered insecticides for cabbage or other leafy greens are effective against diamondbacks, the pests can be controlled in cabbage on Georgia farms. Various formulations of Bacillus thuringiensis (BT), occasionally tank-mixed with certain organophosphate insecticides, have had continued success. On-farm tests have consistently yielded 85 percent or more marketable cabbage where these compounds have been used with the following basic practices:
- use of specially designed high pressure/high volume application equipment;
- application of insecticides at delivery speeds of no more than 4 mph;
- early, close-interval applications (every five days or less) of BT;
- the addition of adequate spreading/sticking agents;
- use of an organophosphate insecticide tank-mixed with BTs only as needed when DBM populations begin to increase;
- avoidance of insecticides that have proven to antagonize efforts to control DBM; and
- pest population monitoring so that other insecticides can be used only when necessary.
Although these measures will not eliminate insecticide resistance, they will help to reverse it. After several years of this method of management, DBM can develop strains that will again be susceptible to currently ineffective insecticides.
Foliage-Feeding Caterpillars
Foliage feeders are the most important pests of cabbage and always pose a serious threat to quality and yield. Even though resistance to insecticides is a major concern, the single most significant problem in control of these pests is the difficulty in maintaining adequate coverage of the plants with insecticidal spray. Most of the eggs of the caterpillar pests are laid either in masses or singly on the underside of the foliage. The larvae, until mature, generally feed on the underside of the foliage or in the bud, making control very difficult.
Another significant problem is that different species of caterpillars are susceptible to different insecticides. When several species are infesting cabbage and greens at the same time, several insecticides may be necessary for adequate control. This increases the cost of production and also creates antagonistic effects in the control of certain pests. Identifying the species complex of a given infestation, maintaining control of the primary species and making judicious insecticide applications for the less significant pest as its population begins to increase are extremely important.
Diamondback moth caterpillar(figures 12, 13, 14, 15) The diamondback moth caterpillar (DBM), Plutella xylostella, is the single most destructive pest to cabbage and leafy greens worldwide. Insecticide resistance has been documented in every corner of the globe; DBM is the key pest in most crucifer cultures. By definition, a key pest is the species whose presence triggers the initial, often early, insecticide applications. These early applications often destroy the natural enemies of both the key pest and secondary pests. Secondary pests may then become economically important. Biorational compounds that are "soft" on natural enemies and provide adequate control of DBM have found a solid niche in our current management strategies. Except for the adult stage, DBM completes its whole life cycle on the plant. DBM moths lay eggs singly on the underside of leaves. The larvae hatch in a day or so and feed on the underside. The larvae grow as large as 5/8 inch. The larvae are green and hang by silken threads when disturbed. They are very active when disturbed. Initial damage is small incomplete holes caused by young larvae and larger complete holes caused by mature larvae. The holes become larger as the leaf develops. The entire plant may become riddled with holes under moderate to heavy populations. Larvae also feed in the developing heads of cabbage, causing deformed heads and encouraging soft rots. The pupae of DBM are green and encased in a netlike cocoon attached to the foliage. Pupae reduce quality as a contaminant. DBM attacks all types of leafy greens and cole crops during all parts of the growing season. DBM is a cold-hardy species, so it can survive cold temperatures in the caterpillar stage. During temperatures below approximately 50°F, larvae cease to feed. As the temperature rises above this mark, feeding resumes. The life cycle is retarded during cooler temperatures. In contrast to this, DBM populations may increase dramatically at temperatures above approximately 80°F. The life cycle may be as long as 50 days at low temperatures and as short as 15 to 20 days during high temperatures. There may be 10 or more generations during warm years. Suppression of DBM populations with preventive treatments is the most efficient control method. Preventive treatments with biological compounds should be made on a five-day interval. A seven-day interval may be used if no worms are found, especially during cool winter weather. Cleanup sprays may be necessary periodically. Heavy rain showers may reduce populations dramatically. Monitor crops two to three times per week and make decisions on changes in control strategies. Note: Transplant beds should be kept free of infestation. |
 Figure 12. Diamondback moth caterpillar (adult). Figure 12. Diamondback moth caterpillar (adult).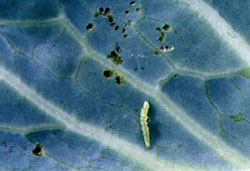 Figure 13. Diamondback moth caterpillar (young larva). Figure 13. Diamondback moth caterpillar (young larva). Figure 14. Diamondback moth caterpillar (mature larva). Figure 14. Diamondback moth caterpillar (mature larva). Figure 15. Diamondback moth caterpillar damage. Figure 15. Diamondback moth caterpillar damage. |
Cabbage Looper(figures 16, 17, 18) The cabbage looper, Trichoplusia ni, is the second most destructive pest to cabbage and leafy greens, and at times is the key pest in Georgia. Biological insecticides are moderately effective, and other insecticides often are needed for adequate control. The cabbage looper is most destructive in early summer and fall. The larvae are large worms (up to 1½ inches) that feed on leaves, creating large holes. Larvae are sluggish and hold on to the plants tenaciously when attempts are made to remove them. This is the only caterpillar pest that has only three pairs of fleshy prolegs near the rear. Except for the adult, the cabbage looper is like the DBM, spending its entire life cycle on the plant. The eggs are laid on the underside of the leaves, and larvae hatch and feed on the underside, with the pupae attached to the underside in a protective cocoon. The pupae are green and two to three times as large as DBM pupae. Controls should be initiated at the first signs of moth activity, whether this is eggs or young larvae. Monitoring the crop two or three times per week helps in making control decisions. |
 Figure 16. Cabbage looper (adult). Figure 16. Cabbage looper (adult). Figure 17. Cabbage looper (larva). Figure 17. Cabbage looper (larva). Figure 18. Cabbage looper (pupa). Figure 18. Cabbage looper (pupa). |
Cabbage Webworm(Figure 19) The cabbage webworm, Hellula rogatalis, is occasionally a serious pest of cabbage, collard and kale. When it occurs, growers are usually caught off guard. Because it has a habit of feeding in the bud area, producing moderate to heavy webbing, growers have difficulty controlling it. Mature larvae are about ¾ inch long and have five dark stripes on a dirty gray body. The head capsule is black with a distinct, white V-shaped mark. Control for the webworm should be initiated at the first signs of an infestation. Some of the same insecticides used against the cabbage looper give good control if coverage in the bud area is excellent. |
 Figure 19. Cabbage webworm. Figure 19. Cabbage webworm. |
Imported Cabbageworm(figures 20, 21) The imported cabbageworm, Pieris rapae, is rarely an economic pest on cabbage and leafy greens if controls for other worms are being applied. The adult is a common butterfly that lays eggs singly on the leaf surface. The larvae are green and have a velvety appearance. Larvae have a narrow, light yellow stripe down the back. Initiate controls when a buildup of larvae occurs. |
 Figure 20. Imported cabbage worm (adult). Figure 20. Imported cabbage worm (adult). Figure 21. Imported cabbage worm (larva). Figure 21. Imported cabbage worm (larva). |
Cross-Striped Cabbageworm(Figure 22) The cross-striped cabbageworm, Evergestis rimosalis, is occasionally a pest in the cooler, northern part of Georgia. It is usually not too difficult to control if the crop is being monitored and timely controls are initiated. The larvae may be slightly longer than ¾ inch and have black and white transverse stripes down the back. Below the transverse stripes on each side is a black and yellow stripe along the length of the body. Initiate controls when larvae are observed. |
 Figure 22. Cross-striped cabbageworm. Figure 22. Cross-striped cabbageworm. |
Beet Armyworm(Figure 23) The beet armyworm, Spodoptera exigua, may be a pest to fall plantings of cabbage, collard, kale, mustard and turnip. Heavy populations that have increased on other crops move to greens crops when food sources become scarce. Diseases during this period often suppress populations below an economic level, but occasionally the beet armyworm can devastate a crop. The beet armyworm is one of the most difficult caterpillars to control. It is naturally resistant to most commonly used insecticides. If it develops into very large populations, control might not be regained. The moth lays masses of eggs on the undersides of leaves. The mass may have up to 150 eggs and is covered with scales off the moth's body, giving the mass a cottony appearance. The larvae are light green to dark olive green and sometimes have stripes of these colors down the back. Above the second pair of legs near the head end is a black spot. Larvae may be 1¼ inches long. Initiate controls if egg masses or larvae are found on 2 percent to 3 percent of the plants. To suppress small populations, apply pesticide every five days. A three- to four-day spray interval may be necessary to bring moderate to heavy populations under control. |
 Figure 23. Beet armyworm. Figure 23. Beet armyworm. |
Corn Earworm(Figure 24) The corn earworm, Helicoverpa zea, formerly Heliothis zea, can be a pest of almost any crop but creates the most serious threat to cabbage. The larvae tunnel into the buds of young plants and the heads of older plants. The larvae are common on many plants and are easily recognized when extracted from the tips of ears of field corn. Make treatments when small larvae are observed on 4 percent to 5 percent of the plants or if large larvae are found on 2 percent or more on the plants. |
 Figure 24. Corn earworm. Figure 24. Corn earworm. |
Cutworm(Figure 25) The granulate cutworm, Feltia subterranea, is the most common species in Georgia's cabbage and leafy greens production areas. Cutworms may survive the winter in the larval stage, so large larvae may be present at the time of planting, especially when planting is made into previous crop residue. Cutworms are recognizable by their greasy dingy gray color and C-shaped posture when at rest. Cutworms feed at night, causing damage to stems and foliage, and retreat into the soil during the day. Treatments for cutworms should be anticipated by inspecting the soil during land preparation so insecticides can be incorporated at planting. |
 Figure 25. Cutworm. Figure 25. Cutworm. |
Other Pests
Seedcorn Maggot(Figure 26) The seedcorn maggot, Delia platura, is a secondary pest that attacks many types of plants. The maggot is a general feeder that is attracted to decaying organic matter. When seedlings are placed under stress, they are most subject to attack by the seedcorn maggot. This occurs most often during the cooler months of planting when transplants are developing slowly. The immature stage is the maggot, which eventually becomes a small housefly-like adult. The maggot damages the plant by entering the roots and stem. Usually, the plant is weakened beyond recovery. Many plants can be killed. The most effective control is to anticipate the conditions that create a favorable environment for maggot attacks and apply the preventive, soil-applied insecticides. Cabbage and collard are the most susceptible to attack. |
 Figure 26. Seedcorn maggots. Figure 26. Seedcorn maggots. |
AphidsSeveral species of aphids attack cabbage and leafy greens. Aphids may be present in fields all year, but they do not always cause significant damage. Aphids are subject to control by several diseases and insect parasites. If broad-spectrum pesticides are used sparingly during the early stages of plant development, aphids usually pose very little threat. However, under conditions that favor rapid development, aphid populations can explode to damaging levels. Cool, dry weather during the spring or fall is ideal for the development of high populations. Cabbage Aphid (Figure 27)The cabbage aphid, Brevicoryne brassicae, is found throughout Georgia. Its appearance differs from other species, with a powdery, waxy covering over its body. Its body is grayish-green. This aphid feeds primarily on cabbage, collard and kale, and seldom feeds on mustard or turnip. The cabbage aphid is difficult to control and should be monitored closely when it is discovered colonizing. Treatments should be made if populations spread beyond the small initial colonies. Turnip AphidThe turnip aphid, Lipaphis erysimi, resembles the cabbage aphid, but lacks a waxy covering and is pale green. The turnip aphid feeds mostly on turnip and mustard. It is difficult to control when conditions favor rapid development. Green Peach Aphid (Figure 28)The green peach aphid, Myzus persicae, is the most common aphid in Georgia and feeds on many vegetable crops and row crops. The wingless types are yellowish-green, green or pink. The winged forms are usually darker. The green peach aphid is most destructive to turnip, mustard, kale and collard but can cause problems in cabbage. Control may be difficult, but can be accomplished with thorough coverage of insecticide sprays. Insecticide controls on seedling stage greens should be avoided until parasites and diseases are given a chance to suppress the population. Turnip Root Aphid (Figure 29)The turnip root aphid, Pemphigus populitransversus, feeds on cabbage, collard, kale, mustard and turnip. Infested plants may be yellow and stunted, but under good growing conditions late infestations often result in very little yield loss. However, turnip roots will be disfigured or discolored and even unmarketable. Preplant incorporation of soil insecticides is the best means of control. Usually this aphid develops higher populations on late fall or early spring plantings. It is difficult to predict, so apply preventive controls only if a history of problems exists. |
 Figure 27. Cabbage aphids. Figure 27. Cabbage aphids. Figure 28. Green peach aphids. Figure 28. Green peach aphids. Figure 29. Turnip root aphids. Figure 29. Turnip root aphids. |
Thrips(Figure 30) Several thrips species feed on cabbage and collard sparingly. Occasionally, damage may be noted. Thrips may be found aggregated in areas damaged by small worms. This behavior is suspected to favor the acquisition of moisture and other nutrients in the exudates of the worm-damaged tissue. Because cabbage is susceptible to tomato spotted wilt virus (TSWV), thrips may become a more important pest in the future. Occasionally, thrips cause a mechanical, "buckskin-type" injury. Controls are not recommended unless heavy populations are observed. |
 Figure 30. Thrips. Figure 30. Thrips. |
Harlequin Bug(figures 31, 32) The harlequin bug, Murgantia histrionica, is rarely a pest of commercial plantings of kale, mustard or turnip. It is more likely to be a problem in cabbage and collard. The harlequin bug is a brightly marked shield-shaped bug that has piercing, sucking mouthparts. It feeds on the veins of leaves, causing the leaf to wilt. Their eggs are barrel-shaped and laid in clusters on the leaves. Eggs are white with two black bands around them. Initiate controls if one bug per 10 plants is found. |
 Figure 31. Harlequin bug (eggs). Figure 31. Harlequin bug (eggs). |
 Figure 32. Harlequin bug (adult). Figure 32. Harlequin bug (adult). |
Stink Bugs(figures 33, 34) Several species of stink bugs attack leafy greens. One of the most common species is the Southern green stink bug, Nezara viridula. Stink bugs commonly infest turnip and mustard more than cabbage and other leafy greens. Stink bugs pierce the plant cell and suck out plant sap. The most common problem with stink bug infestations is that they are a contaminant in processed greens. Control stink bugs when wilting is observed from feeding or when they are found above the accepted threshold for processing. |
 Figure 33. Stink bug (eggs). Figure 33. Stink bug (eggs). Figure 34. Stink bugs (nymphs and adults). Figure 34. Stink bugs (nymphs and adults). |
Chinch Bug(Figure 35) The chinch bug, Blissus leucopterus, may infest turnip and mustard crops, especially when they are planted near corn or small grains. Chinch bugs are small sucking bugs that prefer to feed on grass crops but may migrate to vegetables when these hosts become unsuitable. Even though the adults have wings, they do not fly. Chinch bugs are difficult to control, so early scouting for chinch bugs migrating from nearby sources is important. Initiate controls if a large migrating population is detected. |
 Figure 35. Chinch bug. Figure 35. Chinch bug. |
False Chinch Bug(Figure 36) The false chinch bug, Nysius raphanus, is a fragile sucking bug that is primarily a pest on turnip and mustard. False chinch bugs may infest fields in large numbers. They damage plants by feeding on the veins on the undersides of the leaves. They inject enzymes during the feeding process, causing a green wilting of the leaf margins. This condition is variable; no information is available on how many bugs cause wilting. Therefore, decisions on control are arbitrary, but heavy infestations should not be left uncontrolled. |
 Figure 36. False chinch bug. Figure 36. False chinch bug. |
Silverleaf Whitefly(Figure 37) The silverleaf whitefly, Bemisia argentifolii, is a sporadic pest of cabbage and leafy greens. The silverleaf whitefly may become a problem in late plantings, but is rarely a problem in spring greens. The adult is smaller than a gnat and is bright white with a yellow head and thoracic region. It is mothlike in appearance and feeds on the undersides of leaves, where it also lays eggs. The larvae hatch and become sessile on the underside of the leaf. The adults fly rapidly from the plant when disturbed. Heavy feeding can result in small yellow spots on the foliage of the tender leafy greens. When on cabbage or collard, the whitefly is more a contaminant than an injurious pest. Control for whiteflies is not recommended unless populations in the area are becoming excessively large or honeydew and/or sooty mold is developing on the foliage. Whitefly control is strictly a judgment decision without threshold guidelines. |
 Figure 37. Silverleaf whitefly. Figure 37. Silverleaf whitefly. |
Vegetable Weevil(Figure 38) The vegetable weevil, Listroderes costirostris, may be a pest of seedling cabbage and leafy greens, especially under the cool growing conditions of the early fall and spring plantings. Adults are about ¼- to 3/8-inch long with a stout snout. They are brownish-gray with two nondescript whitish marks on the wing covers. The larvae are white legless grubs. The adult weevil and grub feed directly on the foliage and stems of greens. They can cause significant stand reductions on young plantings. If weevils or grubs are found feeding, apply treatments if more than 5 percent of the stand is being damaged. |
 Figure 38. Vegetable weevil (larvae). Figure 38. Vegetable weevil (larvae). |
Yellow-margined Leaf Beetle(figures 39, 40) The yellow-margined leaf beetle, Microtheca ochroloma, is a small beetle that infests turnip and mustard, especially at field margins. The beetle is black with dirty, yellow-margined wing covers. The larvae are black and alligator-shaped with three pairs of stocky legs. The larvae and adults feed all over the leaves, leaving them with a laced appearance. The pupae may be found in white, round and loosely woven cocoons near the crown of the plants. Initiate controls when larvae and adults are causing noticeable damage and are still present in the field. |
 Figure 39. Yellow-margined leaf beetle (adult). Figure 39. Yellow-margined leaf beetle (adult). |
 Figure 40. Yellow-margined leaf beetle (pupa). Figure 40. Yellow-margined leaf beetle (pupa). |
Weed Management
Managing weeds in leafy greens and cabbage is an important component of overall crop production. With the increased emphasis on integrated pest management and reduced pesticide inputs, weed management efforts will continue to intensify.
Weeds compete with the crop for light, water, nutrients and physical space. In addition, weeds can harbor harmful insects and diseases that can severely damage the present or proceeding crop. This is especially important with crucifer crops such as greens and cole crops, because many common weeds can harbor black-rot (Xanthomonas campestris). This disease is a serious problem for many cole crops. Wild radish can also harbor and increase problems with diamondback moth caterpillars (Plutella xylostella). Weeds also interfere with the harvesting process, either through decreased efficiency with the hand-harvested crops or contamination of machine-harvested commodities. In the case of leafy greens for processing, weed contamination may make the product unmarketable.
As alluded to previously, several weedy species commonly infest cabbage and leafy greens. Weed spectrum is highly influenced by the growing season — whether fall, winter or spring. In the fall, the weed spectrum will consist of summer annual weeds such as crabgrass, pigweed, Florida pusley, Texas panicum, sicklepod and nutsedge. In areas heavily infested with nutsedge, crop production should be avoided. During winter production, weeds such as cutleaf evening primrose, swinecress, Virginia pepperweed, shepherd's purse, henbit, chickweed and wild radish will begin to germinate and grow. These weeds are nearly impossible to control post-emergence in leafy greens and heavy infestations should be avoided, especially areas with wild radish. Spring production generally occurs with decreasing winter weed populations followed by emergence of summer annuals as the temperature increases.
Cultural weed management practices are the most effective methods of weed control because of the lack of chemical weed control alternatives. Such practices include rotation and the stale seedbed technique. The first step in cultural weed management is growing a good crop. Proper fertilization as well as good disease, insect and nematode management are critical. Most leafy greens and cabbage are not highly competitive and early season weed control is necessary for these crops to gain a competitive advantage.
Rotation will continue to be one of the vital components to a successful weed management program in cabbage and leafy greens. Rotation also benefits crop production by lowering the levels of insects and soilborne pathogens and maintains better soil tilth. From a rotational standpoint, controlling weeds in one crop is easier than in another because of: 1) the ability to cultivate, 2) the ability to use different herbicides and 3) the economics of hand weeding. One problem with leafy greens production is the inability to cultivate; thereby, rotational crops that can employ cultivation will help to reduce weed levels.
The stale seedbed technique employs the use of a nonselective herbicide such as paraquat or glyphosate to kill emerged weeds before planting the crop. In the stale seedbed method, the seedbed is prepared as usual but the crop is not planted. The weeds are allowed to emerge and are then killed by the nonselective herbicide. The crop is planted with minimal soil disturbance to prevent stimulation of weed germination. This method can also be used to kill weeds that emerge before the crop. However, great care must be taken to properly identify seedling weeds vs. the crop. This generally works well with weeds such as nutsedge, which commonly emerges three to four days before most crops. Another, nonchemical, method involves reworking the desired area several times before planting, destroying weed seedlings as they emerge. This method seeks to reduce the levels of infestation. Coupled with chemical control just prior to planting, it is an effective means of reducing weed problems.
Mechanical weed control has been, and will continue to be, one of the primary methods of weed control for cole crops such as cabbage, cauliflower, broccoli or collard. Cultivation works extremely well for transplanted crops where small weeds can be eliminated without damaging the crop. Care must be taken to avoid root pruning and excessive leaf damage. If the crop is direct seeded, care must be taken to avoid covering the crop with soil.
Carryover from herbicides is a critical issue with many vegetable crops, especially leafy greens. Leafy greens such as turnip and mustard are very sensitive to many soil active herbicides and great care must be taken to avoid such possibilities. Be sure to know the cropping history of the area, including herbicide use. Rotational restrictions are listed on all herbicide labels and must be followed to avoid severe injury or crop death.
Herbicides approved for use in leafy greens and cabbage are listed in the Georgia Pest Management Handbook. The use of unregistered herbicides is illegal and could result in crop injury and undesirable residues.
Sprayers
Two types of sprayers, boom and air-assisted, are used for applying insecticides, fungicides, herbicides and foliar fertilizers. Air-assisted sprayers (Figure 41) utilize a conventional hydraulic nozzle. Air forces the spray into the plant foliage. Boom sprayers (Figure 42) get their name from the arrangement of the conduit that carries the spray liquid to the nozzles. Booms or long arms on the sprayer extend across a given width to cover a swath as the sprayer passes over the field.
 Figure 41. Air-assisted sprayer. Figure 41. Air-assisted sprayer. |
 Figure 42. Hydraulic boom sprayer. Figure 42. Hydraulic boom sprayer. |
Pumps
Three factors to consider in selecting the proper pump for a sprayer are:
-
Capacity.
The pump should have sufficient capacity to supply the boom output and to provide for agitation (5 to 7 gallons per minute (gpm) per 100-gallon tank capacity). Boom output will vary depending upon the number and size of nozzles. Also, 20 percent to 30 percent should be allowed for pump wear when determining pump capacity. Pump capacities are given in gallons per minute.
-
Pressure.
The pump must produce the desired operating pressure for the spraying job. Pressures are indicated as pounds per square inch (psi).
-
Resistance to corrosion and wear.
The pump must be able to withstand the chemical spray materials without excessive corrosion or wear. Use care in selecting a pump if wettable powders are to be used because these materials will cause pump wear.
Before selecting a pump, consider factors such as cost, service, operating speeds, flow rate, pressure and wear. For spraying vegetable crops, a diaphragm pump is preferred because of serviceability and pressures required.
Nozzles
Nozzle tips are the most neglected and abused part of the sprayer. Because clogging can occur when spraying, clean and test nozzle tips and strainers before each application. When applying chemicals, maintain proper ground speed, boom height and operating pressure. This will ensure proper delivery of the recommended amount of pesticide to the plant canopy.
Herbicides
The type of nozzle used for applying herbicides is one that develops a large droplet and has no drift. The nozzles used for broadcast applications include the extended range flat fan, drift reduction flat fan, turbo flat fan, flooding fan, turbo flooding fan, turbo drop flat fan and wide angle cone nozzles. Operating pressures should be 20 to 30 psi for all except drift reduction and turbo drop flat fans, flooding fans and wide angle cones. Spray pressure more than 40 psi will create significant drift with flat fans nozzles. Drift reduction and turbo drop nozzles should be operated at 40 psi. Flooding fan and wide angle cone nozzles should be operated at 15 to 18 psi. These nozzles will achieve uniform application of the chemical if they are uniformly spaced along the boom. Flat fan nozzles should overlap 50 percent to 60 percent.
Insecticides and Fungicides
Hollow cone nozzles are used primarily for plant foliage penetration for effective insect and disease control and when drift is not a major concern. At pressures of 60 to 200 psi, these nozzles produce small droplets that penetrate plant canopies and cover the undersides of the leaves more effectively than any other nozzle type. The hollow cone nozzles produce a cone-shaped pattern with the spray concentrated in a ring around the outer edge of the pattern. Even fan and hollow cone nozzles can be used for banding insecticide or fungicides over the row.
Nozzle Material
Various types of nozzle bodies and caps, including color-coded versions and multiple-nozzle bodies are available. Nozzle tips are interchangeable and are available in a wide variety of materials, including hardened stainless steel, stainless steel, brass, ceramic, and various types of plastic. Hardened stainless steel and ceramic are the most wear-resistant materials. Use stainless steel tips, with corrosive or abrasive materials, because they have excellent wear resistance. Plastic tips are resistant to corrosion and abrasion and are proving to be very economical for applying pesticides. Brass tips have been common but wear rapidly when used to apply abrasive materials such as wettable powders. Brass tips are economical for limited use, but other types should be considered for more extensive use.
Nozzle Arrangements
When applying insecticides and fungicides, it is advantageous to completely cover both sides of all leaves with spray. When spraying greens, use one nozzle over the top of the row (up to 12 inches wide). Then as the plants start to bush, place nozzles on 10- to 12-inch centers for broadcast spraying. For cabbage, the nozzle arrangement should be adapted for the various growth stages of plants (figures 43 and 44). When plants are small (up to 12 inches) one or two nozzles over the top are sufficient. Then as the plant starts to form a head, drop nozzles should be added in pairs.
This will guarantee that the spray is applied from all directions into the canopy. As the plant increases in height, add additional nozzles for every 8 to 10 inches of growth. In all spray configurations, the nozzle tips should be 6 to 10 inches from the foliage. Properly selected nozzles should be able to apply 25 to 125 gallons per acre when operating at a pressure of 60 to 200 or higher psi. Usually, more than one size of nozzle will be needed to carry out a season-long spray program. Volume and pressure will vary with growth stage and type of material sprayed.
 Figure 43. Use one or two nozzles over the row for small plants. Figure 43. Use one or two nozzles over the row for small plants. |
 Figure 44. Use drop nozzles as plants grow. Figure 44. Use drop nozzles as plants grow. |
Spray Volume - Water Rates (GPA)
The grower who plans to use spray materials at the low water rates should follow all recommendations carefully. Use product label recommendations on water rates to achieve optimal performance. Plant size and condition influence the water rate applied per acre. Examination of the crop behind the sprayer before the spray dries will give a good indication of coverage.
Agitation
Most materials applied by a sprayer are in a mixture or suspension. Uniform application requires a homogeneous solution provided by proper agitation (mixing). The agitation may be produced by jet agitators, volume boosters (sometimes referred to as hydraulic agitators) and mechanical agitators. These can be purchased separately and installed on sprayers. Continuous agitation is needed when applying pesticides that tend to settle out, even when moving from field to field or when stopping for a few minutes.
The procedure below is based on spraying 1/128 of an acre per nozzle or row spacing and collecting the spray that would be released during the time it takes to spray the area. Because 1 gallon contains 128 ounces of liquid, this convenient relationship results in ounces of liquid collected being directly equal to the application rate in gallons per acre.
Calibrate with clean water when applying toxic pesticides mixed with large volumes of water. Check uniformity of nozzle output across the boom. Collect from each for a known time period. Each nozzle should be within 10 percent of the average output. Replace with new nozzles if necessary. When applying materials that are appreciably different from water in weight or flow characteristics, such as fertilizer solutions, etc., calibrate with the material to be applied. Exercise extreme care and use protective equipment when active ingredient is involved.
- From Table 9, determine the distance to drive in the field (two or more runs suggested). For broadcast spraying, measure the distance between nozzles. For band spraying, use band width. For over-the-row or directed spraying, use row spacing.
- Measure the time (seconds) to drive the required distance with all equipment attached and operating. Maintain this throttle setting!
- With the sprayer sitting still and operating at the same throttle setting or engine RPM as used in Step 2, adjust the pressure to the desired setting. The machine must be operated at the same pressure used for calibration.
- For broadcast application, collect spray from one nozzle or outlet for the number of seconds required to travel the calibration distance.
For band application, collect spray from all nozzles or outlets used on one band width for the number of seconds required to travel the calibration distance.
For row application, collect spray from all outlets (nozzles, etc.) used for one row for the number of seconds required to travel the calibration distance. - Measure the amount of liquid collected in fluid ounces. The number of ounces collected is the gallons per acre rate on the coverage basis indicated. For example, if you collect 18 ounces, the sprayer will apply 18 gallons per acre. Adjust applicator speed, pressure, nozzle size, etc., to obtain recommended rate. If speed is adjusted, start at Step 2 and recalibrate. If pressure or nozzles are changed, start at Step 3 and recalibrate.
Table 9. Distance to measure to spray 1/128 acre
One ounce discharged equals one gallon per acre.
| Nozzle Spacing (inches) | Distance (feet) |
| 6 | 681 |
| 8 | 510 |
| 10 | 408 |
| 12 | 340 |
| 14 | 292 |
| 16 | 255 |
| 18 | 227 |
| 20 | 204 |
| 22 | 186 |
| 24 | 170 |
| 30 | 136 |
| 36 | 113 |
| 38 | 107 |
| 40 | 102 |
To determine a calibration distance for an unlisted spacing, divide the spacing expressed in feet into 340.
Example: Calibration distance for a 13" band = 340 ÷ 13/12 = 313 feet.
Sprayers should be calibrated at 2½ to 4 miles per hour. Calibration should be conducted every eight to 10 hours of operation to ensure proper pesticide application.
Irrigation
Even though cabbage and leafy greens are primarily cool-season crops, irrigation will significantly increase their yield and quality in most years. These crops are shallow rooted, and even though their water requirements are less than those for most crops, they can use up the available moisture in the shallow root zone very quickly. The most serious yield reductions result when moisture deficits occur during late development and, in the case of cabbage, during head formation.
Sprinkler irrigation is the only method that has proven practical for irrigation of these crops in Georgia. Common types of systems include center pivot, linear move, traveling big-gun, permanent set and portable aluminum pipe with sprinklers. Each of these systems is satisfactory if used correctly. There are, however, significant differences in initial costs, fuel costs and labor requirements.
Any sprinkler system used on cabbage or greens should be capable of delivering at least 1¼ inches of water each week. In addition, the system should apply the water slowly enough to prevent run-off. With most soils, a rate less than 2 inches per hour safely prevents runoff. Drip irrigation is often used when cabbage, collard or kale are double cropped with another vegetable on mulched beds.
Scheduling Irrigation
The water used by a crop and the water evaporated from the soil is commonly referred to as evapotranspiration (ET). ET rates for cabbage and greens rarely exceed 0.15 inch per day. Factors that affect ET are the stage of crop growth, temperature, relative humidity, solar radiation, wind velocity and plant spacing.
Plant seeded crops into moist soil and irrigate frequently with light applications until germination occurs. If possible, apply 0.25 inch every other day to ensure complete germination. The soil should not become waterlogged.
Plant transplants into moist soil and irrigate with 0.3 to 0.5 inch immediately. This helps to ensure good contact between the soil and roots.
Once a root system is established, maintain soil moisture to a depth of 12 inches. The sandier soils in South Georgia have an available water-holding capacity of about 1 inch per foot of soil depth. Clay soils will hold up to 2 inches per foot. No more than 50 percent of the available water should be depleted before irrigating. This means that net irrigation amounts should be between 0.5 and 1.0 inch per irrigation. The actual amount applied should be 10 percent to 20 percent higher to account for evaporation losses and wind drift. The irrigation frequency will depend on the daily ET rate. In general, during peak water-use periods, sandy soils need 0.6 inch twice a week, and clay soils need 1.2 inches once a week.
Irrigation can best be managed by monitoring the amount of moisture in the soil. Tensiometers or resistance blocks can be used to measure soil moisture. For best results on cabbage and greens, maintain soil tension below 30 centibars. For cabbage, maintaining uniform moisture during head formation is especially important to prevent bursting. Maintain soil moisture until harvest.
Harvest and Handling
Harvesting
 Figure 45. Damaged or diseased leaves should be removed before field packing.
Figure 45. Damaged or diseased leaves should be removed before field packing.Many leafy greens (including cabbage, collard, kale, mustard and turnip) are cut by hand and packed directly in the field for the fresh market. Necessary trimming to remove any yellowed, brownish or damaged leaves should be done as the plants are picked and before they are tied into bunches and placed into containers. In addition, cabbage may be cut by hand, loaded into a bulk container such as a field wagon and hauled to a packing shed for trimming, grading and packaging. During cabbage harvest, cut stems so that they do not extend more than ½ inch beyond the point of attachment of the outmost leaves. Heads may be damaged by excessively long, protruding stems. Bunch collards according to uniform size. Coarse, tough stems of plants should not be packed. Harvest leaves of turnip, mustard and kale when tender by feel, and avoid those showing tough stems.
Ensuring a quality pack can be a problem for hand harvesters. Field labor must be adequately trained and supervised to harvest only optimum maturity and/or sized leaves or rooted plants to meet potential buyer's quality standards.
Field sanitation is very important to reduce the spread of disease among plants. Cutting tools are a primary source of disease carryover. Knives should be routinely sanitized to keep disease inoculum from building on their surfaces and infecting sound cabbage heads or leafy greens. Worker's knives should be collected at the end of a harvest day and placed in a bucket of sanitizer (use 1 ounce of household bleach per gallon of water). For better protection, place buckets of sanitizer at the end of selected rows in the field so workers can sanitize their knives at regular intervals to reduce disease build-up over the course of the production day.
Postharvest Handling/Cooling
When harvesting cabbage or other leafy greens, field crews should exercise care to minimize bruise damage and leaf punctures. Cabbage is sometimes considered a "hardware" item, because it is thrown into bulk containers in the field or at the packing shed. Outer leaves are broken and heads burst when subjected to impact damage. Leaves of leafy greens are crushed if they are overpacked into field boxes. If not used properly, cutting tools will puncture leaves. Any cuts or breaks in the leaves or heads will cause excessive wilting and provide avenues for decay pathogens.
 Figure 46. Pitching cabbage into field wagons or packing line bins causes splitting of heads. Figure 46. Pitching cabbage into field wagons or packing line bins causes splitting of heads. |
 Figure 47. Harvested greens should be shaded from direct sunlight to preserve quality. Figure 47. Harvested greens should be shaded from direct sunlight to preserve quality. |
Cabbage and other greens wilt quickly when there is a delay in removing them from the sun. Leafy greens should be harvested during the coolest part of the day to minimize shriveling and field heat accumulation. If delays occur during packing, shade greens from direct sunlight.
Leafy greens should be cleaned before marketing. Bunches of collards and leaves of mustard, turnip and kale tied in half-dozen bundles are laid on a flatbed trailer and hauled from the field station. A straight-line packing belt conveys bunches beneath spray washers where greens are cleaned to remove sand and dirt and refreshed to improve their appearance. Workers place bundles coming off the end of the belt onto racks and into a storage cooler. Direct field packing of boxed leaves is also done without washing at the request of the buyer.
Quality maintenance is best achieved for leafy greens if they are precooled before shipping. Field workers should trim loose leaves from cabbage during harvest because these leaves interfere with cooling. Buyers prefer three to six wrapper leaves to remain. Packing line graders will typically remove any yellowed, insect damaged or disease damaged heads before boxing. Boxes should have structural integrity (use only new boxes) to prevent product crushing during stacking, loading and distribution. Cabbage is best cooled by vacuum or forced air because rapid air movement is needed to remove heat from solid heads. However, most cabbage packed in Georgia is placed in a room cooler. Other leafy greens (collard, kale, mustard and turnip) should be rapidly cooled using one of the methods above. In addition, most buyers require icing for these greens to provide needed moisture for freshness and crispness. After the field boxing of leaves, they may be taken to the shipping location where a shovel of ice is added to greens in each box. Washed bunches of greens are removed from cooling racks and bulk loaded into trucks by being laid in rows, with top icing for each row. Icing takes 2.2 pounds of ice for every 4 pounds of greens to maintain the temperature below 40°F. All leafy greens, including cabbage, should be cooled to 32°F before marketing.
Quality Grade Standards
Cabbage
U.S. No.1 and U.S. Commercial are the two U.S. grade standards provided.
U.S. No.1 consists of heads of cabbage of one cultivar or similar varietal characteristics. The heads must be reasonably solid; not withered, puffy or burst; and free of soft rot, seedstems and damage caused by discoloration, freezing, disease, insects, mechanical or other means. Each head shall be well trimmed.
U.S. Commercial consists of heads of cabbage that meet requirements for U.S. No.1 grade, but it has an greater tolerance for defects and allows heads to be reasonably firm rather than solid. A minimum size or minimum and maximum sizes may be specified in connection with the grade, such as "U.S. No.1, one pound minimum" or "U.S. No.1, two to four pounds." To allow for variations incident to proper grading and handling, defect tolerances based on product weight are provided in Table 10.
Collard
U.S. standards for collard greens provide for one grade: U.S. No.1. This consists of greens of similar varietal characteristics that are fresh, fairly tender, fairly clean, well trimmed and of characteristic color for the variety or type. Also, greens shall be free from decay, damage caused by coarse stems and seedstems, discoloration, freezing, foreign material, disease, insects, mechanical or other means. Tolerances for grading collard greens are based on product weight in a container (see Table 10 for defect percentages).
Kale
U.S. Standards for kale provide two grades: U.S. No.1 and U.S. Commercial. Buyers customarily use U.S. No.1, which requires plants to be of one type: well trimmed, not stunted, free of decay and of damage caused by yellow or discolored leaves, seedstems, wilting, bud burn, freezing, dirt, disease, insects, mechanical or other means. Tolerances for grading kale are based on product weight in a container (see Table 10).
Mustard and Turnip
U.S. Standards for mustard greens and turnip greens provide for one grade: U.S. No. 1. This consists of greens of similar varietal characteristics that are fresh, fairly tender, fairly clean and free of decay and of damage caused by seedstems, discoloration, freezing, foreign material, disease, insects, mechanical or other means. To allow for variations incident to proper handling and grading, defect tolerances for each are provided based on weight (see Table 10).
Table 10. U.S. Grade standards based on allowable defect levels
| Commodity | Grade | Tolerances by Weight |
Size Requirement | Trim Requirement |
| Cabbage | U.S. No.1 | 10% total, including 2% soft decay | For off-size: 15% total, but not more than 10% above or below size |
For trimming: 10% may fail to meet required number of wrapper leaves |
| U.S. Commercial |
25% total, including 10% serious damage and 2% soft decay | |||
| Kale | U.S. No.1 | 10% total, including 1% wet decay | Size not a requirement of grade |
Kale must be well trimmed |
| Collard, Mustard, Turnip |
U.S. No.1 | 10% total, including 5% serious damage and 2% decay | Size reported as small, medium or large leaves | Mustard and turnip greens have no trimming requirements. Collard must be well trimmed. |
Note: Some buyers expect higher quality than these limits.
 Figure 48. Damaged or used containers allow product crushing during distribution and bring lower prices. Figure 48. Damaged or used containers allow product crushing during distribution and bring lower prices. |
 Figure 49. It takes approximately 2 pounds of ice for every 4 pounds of leafy greens to maintain freshness and crispness. Figure 49. It takes approximately 2 pounds of ice for every 4 pounds of leafy greens to maintain freshness and crispness. |
Packaging
Cabbage is packed in 1.8 bushel (50 pound) waxed, corrugated cartons, 1.75 bushel crate or large meshed bags in Georgia. Proper sizing and count per box are important for marketing. Buyers demand uniformly sized heads, 18 to 22 count, in cartons. Cartons bring a premium price and help protect the heads from damage during distribution. Larger heads, 10 to 14 count, are packed in meshed bags and bring a lower price.
The most common package for Georgia greens is the 1 3/5 bushel crate, which is shipped mostly to the northeastern markets. The 13/5 crate generally holds 16 to 18 bunches. Shipments to Midwestern markets are usually in a standard Western carton, which holds 24 bunches. Loose greens are packed in a carton and are usually shipped to markets in the Southeast. Each container holds about 25 pounds. Specifically, greens are packaged as described below.
Collards are sold in bunches of two or three plants. Collards are bulk loaded into trucks or packed into wirebound boxes. Depending on market demand, bunches are packed 12 to 24 bunches per box or sold by the dozen. Collards are top iced for sale.
Kale is stripped from the plant and tied into bunches for marketing. Usually, 12 to 16 leaves compose a bunch and bundled leaves are sold as half-dozen bundles per box. Kale is iced before sale.
Mustard greens are marketed as bundled leaves. Freshness and lack of wilt are important marketing factors. Bundles of 12 to 16 leaves are packed into wirebound or waxed cartons (18 to 20 pounds, excluding ice) depending on market demand. Leaves are iced before sale.
Turnip greens are sold as rooted plants and leaves. If marketed as plants, they should be harvested when the roots reach 1½ to 2 inches in diameter and bunched as two or three plants, similar to collard. Depending on market demand, turnips are packed into 25- or 50-pound cartons or by the dozen as bunches in wirebound crates. Turnip leaves are tied into bundles and sold, with half-dozen bundles per box. Rooted plants and leaves are top iced during packaging.
Storage/Shipping
Maximum storage time for cabbage is three to six weeks if it has been properly precooled and held at 32°F. A relative humidity of 90 percent to 95 percent should be maintained during storage. Cabbage is compatible for holding and shipment with fruits and vegetables that do not produce the ripening gas ethylene. Quality is damaged by exposure to trace amounts of ethylene. Yellowing and shedding of outer leaves results. Do not store or ship cabbage with ripening tomatoes, cantaloupes or ethylene-producing fruits such as peaches.
Other leafy greens (collard, kale, mustard and turnip) require the same storage conditions (temperature, 32°F; humidity, 95 percent) but have a shelf life of only two weeks. This is due to their higher respiration rate and leafy nature, which causes them to lose moisture rapidly. In addition to cold storage, greens should be top iced to retain crispness. These greens are also ethylene sensitive and should not be stored or shipped with ripening tomatoes, cantaloupes or fruits such as peaches.
Production Costs
Cabbage and greens growers can use enterprise budgets to estimate production and break-even prices. Budgets include cost estimates for those inputs necessary to achieve the specified yields over a period of years. Production practices vary among growers, so each grower should adapt budget estimates to reflect the individual situation. Detailed printed and computerized budgets are available from your county Extension agent.
Types of Costs
The total cost of producing any crop includes variable and fixed costs. The variable (operating) costs vary with the cultural practices used. Common variable costs include seed, fertilizer, chemicals, fuel and labor. Fixed costs include items such as equipment ownership (deprecation, interest, insurance and taxes), management and general overhead costs. Most of these costs are incurred even if little production takes place.
Variable costs are further broken down into preharvest operations in the budgets. This provides an opportunity to analyze the costs at different stages of the production process.
Land cost can be a variable or a fixed cost. Even if you own the land, cost is involved. Land is fixed cost in the sample budget. If land is double-cropped, charge each enterprise half the annual cost.
A fixed cost per hour of use shows ownership costs for tractors and equipment (depreciation, interest, taxes, insurance and shelter). Overhead and management are 15 percent of all pre-harvest variable expenses. This figure pays for management and farm costs that cannot be allocated to any one specific enterprise. Overhead items include utilities, farm shop and equipment, pick-up trucks and fees.
Cost per Unit of Production
The cost categories (tables 11 and 12) are broken down by cost per unit at the bottom of the budget. The preharvest variable costs and the fixed costs decline with increases in yields.
| Costs per carton of cabbage for 1998 are estimated to be: | |
| Preharvest Cost |
$1.54
|
| Harvest and Marketing Cost |
$2.39
|
| Fixed Cost |
$ .54
|
| Total Cost |
$4.47
|
|
Costs per carton of leafy greens for 1998 are estimated to be: |
|
| Preharvest Cost |
$1.03
|
| Harvest and Marketing Cost |
$2.95
|
| Fixed Cost |
$ .46
|
| Total Cost |
$4.44
|
For current cost estimates, see the most recent Extension vegetable budgets, available from your county Extension agent.
Budget Uses
In addition to estimating the total costs and break-even prices for producing cabbage and greens, the budgets have other uses.
Estimates of the cash costs (out-of-pocket expenses) provide information on how much money must be borrowed. The cash cost estimates are helpful in preparing cash flow statements.
When growers use share leases, the cost estimates by item can be used to determine more accurately a fair share arrangement by the landlord and tenant.
Risk-Rated Net Returns
Because yields and prices vary from year to year, an attempt is made to estimate the riskiness of producing cabbage and greens. The Agricultural and Applied Economics Department uses five yields and prices to calculate risk. The median values are those prices and yields a particular grower would anticipate exceeding half the time; half the time, he would anticipate not reaching these prices and yields. Optimistic values are those prices and yields a grower would expect to reach or exceed one year in six. The pessimistic values are poor prices and yields that would be expected one year in six. The best and worst values are those extreme levels that would occur once in a lifetime (one in 48).
The risk-rated section for cabbage (Table 13) shows a 69 percent chance of covering all costs. Over a period of years, this hypothetical grower would anticipate an average or expected return of $479 per acre. One year out of six, he would expect to make more than $1,459 per acre or to lose more than $526 per acre.
The risk-rated section (Table 14) for leafy greens shows a 54 percent chance of covering all costs. Over a period of years, this hypothetical grower would anticipate an average or expected returns of $38 per acre. He would be expected to net $38 or more about half the time and net $38 or less half the time. One year out of six he would expect to make more than $490 per acre or to lose more than $426 per acre.
Table 11. Estimated cabbage yields, prices and costs
|
Best
|
Optimistic
|
Median
|
Pessimistic
|
Worst
|
|
| Yield (cartons) |
900
|
700
|
500
|
300
|
0
|
| Price per carton |
8.50
|
7.00
|
5.50
|
4.00
|
2.50
|
| Item |
Unit
|
Quantity
|
Price
|
Dollar
Amount per Acre |
| Variable Costs | ||||
|
1,000
|
16.00
|
26.00
|
416.00
|
|
|
Lime, Applied |
Ton
|
.50
|
26.00
|
13.00
|
|
Fertilizer |
Cwt
|
12.00
|
8.50
|
102.00
|
|
Sidedressing |
Acre
|
1.00
|
22.80
|
22.80
|
|
Insecticide |
Application
|
7.00
|
4.50
|
31.50
|
|
Fungicide |
Application
|
10.00
|
2.90
|
29.00
|
|
Herbicide |
Acre
|
1.00
|
13.50
|
13.50
|
|
Machinery |
Hour
|
6.00
|
5.50
|
33.00
|
|
Labor |
Hour
|
10.00
|
6.00
|
60.00
|
|
Irrigation |
Application
|
3.00
|
4.26
|
12.78
|
|
Interest on Operating Capital |
Dollar
|
733.58
|
10.5%
|
38.51
|
|
Preharvest Variable Costs |
772.09
|
|||
|
Harvest and Marketing Costs |
||||
|
Labor |
Hours
|
50
|
5.25
|
262.50
|
|
Container |
Carton
|
500
|
1.30
|
650.00
|
|
Hauling & Marketing |
Carton
|
500
|
.57
|
285.00
|
|
Total Harvest and Marketing |
1,197.50
|
|||
|
Total Variable Costs |
1,969.59
|
|||
|
Fixed Costs |
||||
|
Machinery |
Hour
|
6.00
|
11.31
|
67.86
|
|
Irrigation |
Acre
|
1.00
|
47.10
|
47.10
|
|
Land |
Acre
|
1.00
|
40.00
|
40.00
|
|
Overhead and Management |
Dollar
|
772.09
|
.15
|
115.81
|
|
Total Fixed Costs |
270.77
|
|||
| Total Budgeted Cost |
2,240.36
|
|||
| Costs Per Carton | ||||
|
Preharvest Variable Cost per Carton |
1.54
|
|||
|
Harvest and Marketing Cost per Carton |
2.39
|
|||
|
Fixed Cost per Carton |
.54
|
|||
|
Total Budgeted Cost per Carton |
4.47
|
|
||
Table 12. Estimated leafy greens yields, prices and costs
|
Best
|
Optimistic
|
Median
|
Pessimistic
|
Worst
|
|
| Yield (cartons) |
600
|
475
|
350
|
200
|
0
|
| Price per carton |
7.00
|
5.90
|
4.75
|
3.60
|
2.5
|
| Item |
Unit
|
Quantity
|
Price
|
Dollar
Amount per Acre |
|
| Variable Costs | |||||
| Seed |
Pounds
|
3.50
|
5.50
|
19.25
|
|
|
Lime, Applied |
Ton
|
.50
|
26.00
|
13.00
|
|
|
Nitrogen |
Pounds
|
180.00
|
.28
|
50.40
|
|
|
Phosphorus |
Pounds
|
200.00
|
.31
|
62.00
|
|
|
Potassium |
Pounds
|
200.00
|
.14
|
28.00
|
|
|
Sulfur |
Pounds
|
10.00
|
.21
|
2.10
|
|
|
Boron |
Pounds
|
1.00
|
2.80
|
2.80
|
|
|
Herbicide |
Acre
|
1.00
|
5.85
|
5.85
|
|
|
Fungicide |
Acre
|
1.00
|
50.00
|
50.00
|
|
|
Insecticide |
Acre
|
1.00
|
27.60
|
27.60
|
|
|
Machinery |
Hour
|
2.00
|
6.90
|
13.80
|
|
|
Labor |
Hour
|
4.00
|
6.00
|
24.00
|
|
|
Irrigation |
Inch
|
10.00
|
4.26
|
42.60
|
|
|
Interest on Operating Capital |
Dollar
|
341.40
|
10.5%
|
17.92
|
|
|
Preharvest Variable Costs |
359.32
|
||||
|
Harvest and Marketing Costs |
|||||
|
Labor |
Carton
|
350
|
1.25
|
437.50
|
|
|
Container |
Carton
|
350
|
1.30
|
455.00
|
|
|
Hauling & Marketing |
Carton
|
350
|
.40
|
140.00
|
|
|
Total Harvest and Marketing |
1,032.50
|
||||
|
Total Variable Costs |
1,391.82
|
||||
|
Fixed Costs |
|||||
|
Machinery |
Hour
|
2.00
|
15.74
|
31.48
|
|
|
Irrigation |
Acre
|
1.00
|
35.00
|
35.00
|
|
|
Land |
Acre
|
1.00
|
40.00
|
40.00
|
|
|
Overhead and Management |
Dollar
|
359.32
|
.15
|
53.90
|
|
|
Total Fixed Costs |
160.38
|
||||
| Total Budgeted Cost |
1,552.20
|
||||
| Costs Per Carton | |||||
|
Preharvest Variable Cost per Carton |
1.03
|
||||
|
Harvest and Marketing Cost per Carton |
2.95
|
||||
|
Fixed Cost per Carton |
.46
|
||||
|
Total Budgeted Cost per Carton |
4.44
|
||||
Table 13. Risk-rated cabbage returns over total costs
|
Optimistic
|
Expected
|
Pessimistic
|
|||||
|
Net Returns ($)
|
1,949
|
1,459
|
969
|
479
|
-23
|
-526
|
-1,028
|
|
Chances of Obtaining This Level or More
|
7%
|
16%
|
31%
|
50%
|
|||
|
Chances of Obtaining This Level or Less
|
50%
|
31%
|
16%
|
7%
|
|||
| Chance for Profit = 69% |
Base Budgeted Net Returns¹ = $511
|
1 Base budgeted net returns are the returns that would be estimated if yields and prices were estimated to be the same each year.
Table 14. Risk-rated leafy greens returns over total costs
|
Optimistic
|
Expected
|
Pessimistic
|
|||||
|
Net Returns ($)
|
715
|
490
|
264
|
38
|
-194
|
-426
|
-659
|
|
Chances of Obtaining This Level or More
|
7%
|
16%
|
31%
|
50%
|
|||
|
Chances of Obtaining This Level or Less
|
50%
|
31%
|
16%
|
7%
|
|||
| Chance for Profit = 54% |
Base Budgeted Net Returns = $118
|
Marketing Cabbage and Leafy Greens
Marketing cabbage, greens or any vegetable is more than selling. Marketing includes production, distribution and pricing. To be successful, marketing must be responsive to consumers' demands. Consumers demand quality, freshness and "reasonable" prices.
Production
Production data for most vegetables are not available. Thus, other types of data must be used to substitute for production estimates. USDA collects market arrival data for "fresh" fruits and vegetables in the major markets in the United States.
During 1996, these data show, 31 states marketed cabbage at some time during the year. The top 10 producing states accounted for about 90 percent of the annual shipments to the major U.S. markets. California was the leader and was followed by Texas, New York and Florida. Georgia ranked fifth in shipments (see Table 15).
Cabbage is available throughout the year, but volume peaks in March. Georgia's share of annual volume has grown from about 4 percent in the mid-1980s to 7.5 percent to 10 percent of the annual volume by the mid-1990s. Georgia volume peaks in May but accounts for more than 10 percent of U.S. volume during December to January and May to June. Florida and Texas are the primary competitors in May; North Carolina is the primary competition in June.
Georgia primarily markets cabbage east of the Mississippi. The South (primarily the Atlanta area) accounts for about two-thirds of Georgia's shipments. Boston, Cincinnati and Chicago are also major markets for Georgia.
Twenty-nine states market greens some time during the year. However, seven states account for more than 80 percent of the annual volume shipped to the 20 major U.S. Markets (see Table 16). California and Georgia are the leading greens producing states. Texas and New Jersey follow in annual volume.
Although greens are available year-round, production peaks during November through May. Georgia's volume peaks during April and May. Georgia accounts for 20 percent to 30 percent of the annual volume but has more than 30 percent of the market in December, April and May.
The top three greens markets are Los Angles, Atlanta and Boston. The top markets for Georgia's greens are Atlanta, Cincinnati and Boston. Cincinnati seems to be a larger greens market than would be expected — receiving nearly the same volume as Chicago. The Cincinnati metropolitan area is the 23rd largest; Chicago is the third. Apparently, Georgia greens growers have developed a niche in the Cincinnati market. Nationally, greens are distributed fairly evenly (24 percent to 26 percent) in the four major geographical areas of the country. However, data for greens are not separated by type of greens, so one area may be consuming a higher proportion of one type when compared with other areas. The Southern cities receive more than 40 percent of Georgia's greens.
Table 15. Average monthly cabbage shipments (million pounds) to 20 U.S. cities, 1996
|
State |
Jan.
|
Feb.
|
March
|
April
|
May
|
June
|
July
|
Aug.
|
Sept.
|
Oct.
|
Nov.
|
Dec.
|
1996
|
1995
|
| California |
7.3
|
6.5
|
8.7
|
8.0
|
7.8
|
7.0
|
6.2
|
6.3
|
5.7
|
6.9
|
6.3
|
6.1
|
82.8
|
84.2
|
| Texas |
13.9
|
11.6
|
17.1
|
14.8
|
11.5
|
2.0
|
0.4
|
0.3
|
0.0
|
0.1
|
1.1
|
3.5
|
76.3
|
59.2
|
| New York |
6.8
|
5.0
|
5.2
|
4.7
|
4.2
|
2.0
|
6.0
|
6.2
|
7.6
|
9.5
|
10.0
|
6.5
|
73.7
|
83.3
|
| Florida |
6.2
|
9.1
|
11.6
|
15.1
|
13.2
|
1.5
|
0.0
|
0.0
|
0.0
|
0.0
|
0.3
|
4.4
|
61.4
|
63.3
|
| Georgia |
4.4
|
1.8
|
0.5
|
0.9
|
7.7
|
5.5
|
3.7
|
1.1
|
1.1
|
1.6
|
1.4
|
6.3
|
36.0
|
45.2
|
| New Jersey |
–
|
–
|
–
|
–
|
–
|
2.4
|
7.1
|
2.4
|
2.9
|
3.1
|
3.9
|
1.5
|
23.3
|
14.1
|
| North Carolina |
0.2
|
–
|
–
|
–
|
1.0
|
7.5
|
2.9
|
1.5
|
0.7
|
1.2
|
1.5
|
1.7
|
18.2
|
19.2
|
| Wisconsin |
–
|
–
|
–
|
–
|
–
|
–
|
0.7
|
3.3
|
3.4
|
4.2
|
3.4
|
0.5
|
15.5
|
21.0
|
| Illinois |
–
|
–
|
–
|
–
|
–
|
0.7
|
2.1
|
2.6
|
2.2
|
2.5
|
1.0
|
0.2
|
11.3
|
10.0
|
| Pennsylvania |
–
|
–
|
–
|
–
|
–
|
–
|
0.7
|
2.9
|
2.9
|
2.4
|
1.7
|
0.4
|
11.0
|
11.6
|
| Subtotal |
38.8
|
34.0
|
43.1
|
43.5
|
45.4
|
28.6
|
29.8
|
26.6
|
26.5
|
31.5
|
30.6
|
31.1
|
409.5
|
411.1
|
| Total |
43.9
|
37.9
|
47.2
|
45.2
|
46.6
|
30.6
|
40.1
|
34.8
|
34.4
|
39.7
|
35.7
|
35.7
|
471.8
|
456.9
|
| Subtotal/Total |
88.4%
|
89.7%
|
91.3%
|
96.2%
|
97.4%
|
93.5%
|
74.3%
|
76.4%
|
77.0%
|
79.3%
|
85.7%
|
87.1%
|
86.8%
|
90.0%
|
|
Georgia/Total |
10.0%
|
4.7%
|
1.1%
|
2.0%
|
16.5%
|
18.0%
|
9.2%
|
3.2%
|
3.2%
|
4.0%
|
3.9%
|
17.6%
|
7.6%
|
9.9%
|
Table 16. Average monthly greens shipments (million pounds) to 20 U.S. Cities, 1996
|
State |
Jan.
|
Feb.
|
March
|
April
|
May
|
June
|
July
|
Aug.
|
Sept.
|
Oct.
|
Nov.
|
Dec.
|
1996
|
1995
|
| California |
4.2
|
4.8
|
4.6
|
5.4
|
5.4
|
4.2
|
4.1
|
4.1
|
3.2
|
4.5
|
4.7
|
4.7
|
53.9
|
42.5
|
| Georgia |
4.8
|
3.7
|
3.3
|
5.5
|
6.2
|
1.9
|
1.5
|
0.6
|
0.7
|
1.8
|
3.0
|
5.6
|
38.6
|
47.5
|
| Texas |
2.7
|
5.1
|
4.8
|
4.2
|
2.2
|
0.0
|
0.0
|
0.0
|
0.0
|
0.1
|
1.0
|
1.7
|
21.8
|
11.2
|
| New Jersey |
–
|
–
|
–
|
0.1
|
1.6
|
2.4
|
3.2
|
1.7
|
1.8
|
1.8
|
1.6
|
0.1
|
14.3
|
12.4
|
| Arizona |
2.4
|
2.5
|
3.1
|
1.6
|
0.2
|
–
|
–
|
–
|
–
|
–
|
0.2
|
0.8
|
10.8
|
5.9
|
| Maryland |
–
|
–
|
–
|
0.1
|
0.1
|
0.7
|
1.0
|
0.9
|
1.0
|
1.0
|
0.9
|
0.1
|
5.8
|
5.9
|
| North Carolina |
0.2
|
0.2
|
0.2
|
0.2
|
1.8
|
0.4
|
0.2
|
0.1
|
0.1
|
0.2
|
0.8
|
0.9
|
5.3
|
7.0
|
| Subtotal |
14.3
|
16.3
|
16.0
|
17.1
|
17.5
|
9.6
|
10.0
|
7.4
|
6.8
|
9.4
|
12.2
|
13.9
|
150.5
|
132.4
|
| Total |
16.5
|
18.1
|
17.6
|
18.3
|
18.8
|
11.5
|
14.5
|
12.5
|
11.6
|
14.0
|
15.2
|
16.2
|
184.8
|
164.4
|
| Subtotal/Total |
86.7%
|
90.1%
|
90.9%
|
93.4%
|
93.1%
|
83.5%
|
69.0%
|
59.2%
|
58.6%
|
67.1%
|
80.3%
|
85.8%
|
81.4%
|
80.5%
|
|
Georgia/Total |
29.1%
|
20.4%
|
18.7%
|
30.1%
|
33.0%
|
16.5%
|
10.3%
|
4.8%
|
6.0%
|
12.9%
|
19.7%
|
34.6%
|
20.9%
|
28.9%
|
Table 17. Average cabbage arrivals in 20 U.S. Cities
| Location |
From Georgia
|
From U.S.
|
||
|
Million Pounds
|
Percent
|
Million Pounds
|
Percent
|
|
| Atlanta |
12.7
|
53%
|
40.2
|
8%
|
| Baltimore-Washington |
1.9
|
8%
|
35.9
|
7%
|
| Columbia |
0.2
|
1%
|
10.5
|
2%
|
| Dallas |
0.1
|
0%
|
25.7
|
5%
|
| Miami |
0.5
|
2%
|
6.7
|
1%
|
| New Orleans |
0.1
|
0%
|
10.5
|
2%
|
| South |
15.5
|
64%
|
129.5
|
25%
|
| Boston |
1.7
|
7%
|
41.8
|
8%
|
| Buffalo |
0.2
|
1%
|
8.6
|
2%
|
| New York-Newark |
1.3
|
5%
|
35.3
|
7%
|
| Philadelphia |
1.0
|
4%
|
27.6
|
5%
|
| Pittsburgh |
0.6
|
3%
|
19.3
|
4%
|
| Northeast |
4.8
|
20%
|
132.6
|
26%
|
| Cincinnati |
1.5
|
6%
|
27.9
|
5%
|
| Chicago |
1.3
|
5%
|
43.4
|
8%
|
| Denver |
8.0
|
2%
|
||
| Detroit |
0.6
|
3%
|
11.4
|
2%
|
| St. Louis |
0.5
|
2%
|
17.8
|
4%
|
| Midwest |
3.9
|
16%
|
108.5
|
21%
|
| Los Angeles |
87.7
|
17%
|
||
| San Francisco |
36.2
|
7%
|
||
| Seattle-Tacoma |
17.7
|
4%
|
||
| West |
141.6
|
28%
|
||
|
Total |
24.2
|
100%
|
512.2
|
100%
|
Table 18. Average greens arrivals in 20 U.S. Cities
| Location |
From Georgia
|
From U.S.
|
||
|
Million Pounds
|
Percent
|
Million Pounds
|
Percent
|
|
| Atlanta |
12.5
|
32%
|
17.1
|
11%
|
| Baltimore-Washington |
2.9
|
8%
|
9.1
|
6%
|
| Columbia |
2.9
|
2%
|
||
| Dallas |
0.3
|
1%
|
8.0
|
5%
|
| Miami |
1.1
|
1%
|
||
| New Orleans |
|
|
1.8
|
1%
|
| South |
15.7
|
41%
|
40.0
|
26%
|
| Boston |
5.1
|
13%
|
15.0
|
9%
|
| Buffalo |
0.1
|
0%
|
1.2
|
1%
|
| New York-Newark |
3.6
|
9%
|
14.8
|
9%
|
| Philadelphia |
2.2
|
6%
|
8.4
|
5%
|
| Pittsburgh |
|
|
0.4
|
0%
|
| Northeast |
11.0
|
28%
|
39.8
|
24%
|
| Cincinnati |
1.7
|
4%
|
14.1
|
9%
|
| Chicago |
9.9
|
26%
|
13.9
|
9%
|
| Denver |
1.7
|
1%
|
||
| Detroit |
0.2
|
0%
|
9.2
|
6%
|
| St. Louis |
0.4
|
1%
|
3.8
|
2%
|
| Midwest |
12.2
|
31%
|
42.7
|
26%
|
| Los Angeles |
25.8
|
16%
|
||
| San Francisco |
8.9
|
5%
|
||
| Seattle |
4.7
|
3%
|
||
| West |
34.9
|
24%
|
||
|
Total |
38.9
|
100%
|
161.9
|
100%
|
Pricing
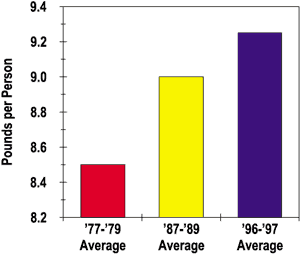 Figure 50. Cabbage consumption.
Figure 50. Cabbage consumption.Supply and demand determine the general price level. The competing states' production determines the supply. Consumers' willingness to buy different quantities at different prices determines the demand.
Consumption data are reported for cabbage but not for greens (see Figure 50). Cabbage and greens prices vary greatly within a season and between years. Weather effects on production cause most of the price variation within season. Changes in acreage and weather cause price variations among years. Demand changes are slight from year-to-year and cause very little of the price variation.
Figure 51 shows the average May price for Georgia cabbage for the past 20 years. Prices have not changed much over the period. During the 1978-87 period, May prices for Georgia cabbage averaged $5.15 per carton, even with the extremely high 1982. During the 1988-97 period, the average price was $6.00. Figure 52 shows the Georgia greens prices for the April and May period. Most years, the prices fluctuated around $4.00 per 25-pound carton. However, the 1996 freeze caused a spike in prices. For recent prices, see Extension agricultural economics publication Vegetable Economics – A Planning Guide.
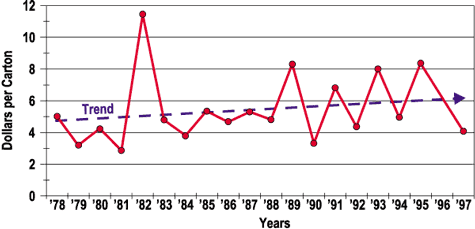 Figure 51. May cabbage prices. Figure 51. May cabbage prices. |
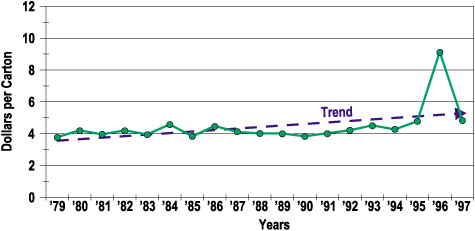 Figure 52. April-May cabbage prices. Figure 52. April-May cabbage prices. |
Summary
Nationally, cabbage is experiencing some growth. Consumption is increasing; prices are trending up. Cabbage seems to be profitable for many growers only when a competing area experiences adverse weather. Greens are experiencing a similar favorable marketing environment. Production seems to be increasing while prices have been fairly stable.
Cabbage and greens growers will have to continue to adjust to changing market conditions. For these highly competitive commodities, the better marketers will be the ones most likely to survive.
Acknowledgments
The authors would like to express their gratitude to the following people without whose help this publication would not have been possible:
- Mrs. Jan Howell, Ag Engineering Department;
- Mrs. Kay Dunn, Horticulture Department;
- Mrs. Soccoro Seela, Entomology Department;
- Mrs. Priscilla Dolney, Ag Engineering Department;
- Mrs. Alice Pitts, Agricultural and Applied Economics Department;
- Mrs. Machelle Clements, Plant Pathology Department;
- Miss Whatiki Crawford, Crop and Soil Science Department; and
- Dr. Johnny Dan Gay, former Extension Plant Pathologist.
Status and Revision History
Published on Oct 01, 2000
Published on Feb 26, 2009
Published with Full Review on Mar 30, 2014
Published with Full Review on Jan 30, 2017
