
- Preface
- Culture
- Soils and Fertilizer Management
- Irrigation
- Disease Management
- Insect Management
- Weed Management
- Pesticide Application
- Good Agricultural Practices in the Harvest, Handling and Packing of Cantaloupes
- Production Costs
- Cantaloupe Marketing
Preface
This publication was compiled to meet the growing cantaloupe industry in Georgia. Its 10 chapters represent the latest information available on successful cantaloupe and specialty melon production. This publication is the compilation of information through the Georgia Vegetable Team, a cross-discipline commodity group within the University of Georgia. Thanks are extended to all the contributors and reviewers for their efforts in putting this publication together.
Culture
Description
Cantaloupe and specialty melons are members of the cucurbit (Cucurbitaceae) family, which also includes several warm season vegetables such as watermelon, squash and cucumber. Cantaloupes and specialty melons grow as prostrate vines with andromonecious flowering, both perfect (with male and female flower parts) and imperfect (male flowers).
The scientific name for cantaloupes (muskmelons) and specialty melons is Cucumis melo. This species is subdivided into seven botanical variants: cantaloupensis, reticulatous, inodorous, flexuosus, conomon, chito and dudaim. Only two of the seven variants have significant commercial importance in the United States. These are the reticulatous and inodorous variants.
Interestingly, the cantaloupensis variant or true cantaloupe is not grown commercially in North America. The fruit of this type is medium sized, warty or scaly and is more commonly found in Europe. Members of the flexuosus group are referred to as snake melons because of their long slender shape. The conomon type composes the oriental pickling melons. Chito melons include the mango and garden lemon melons. The dudaim variant includes the pomegranate or Queen Anne?s pocket melon. None of these groups is of much economic importance in the United States, although occasionally they are grown for local markets.
The reticulatous variant is the most commonly grown in the United States and includes the cantaloupe or muskmelon. Several different types of melons are among this group. The terms "muskmelon" and "cantaloupe" are used interchangeably in this country, with the term "cantaloupe" used more widely.
Cantaloupes or muskmelons can be placed into several different categories based on fruit type.
The terms "western" or "shipping" are used for cantaloupes that have uniformly netted rinds, orange flesh and lack any sutures. Traditionally this type was grown in western states and shipped throughout the country. They are, however, widely adapted and grown throughout the United States. They can be harvested when sufficiently sized but not fully mature. The stem at this stage will partially pull free. This is referred to as the half-slip stage. These fruit, protected by the netted rind and still immature, ship easily.
Conversely, the terms "eastern" and "jumbo" are used to describe melons that have been traditionally grown for local markets. These melons usually have less uniform or no netting with deep sutures. They are often larger and less uniform in size than western cantaloupes. The flesh of these melons is orange or salmon colored. Some people reserve the term "muskmelon" for this type of melon.
Other less widely grown cantaloupes include Galia, Persian and Charentais. Galia types have netted rinds like typical cantaloupes but with green flesh like honeydews. Charentais have smooth rinds with sutures. The rind color is usually gray or gray-blue in color. The flesh is orange. Galia and Charentais both slip from the vine at maturity. Persian melons are larger than cantaloupes with dark green rinds covered with fine netting. The flesh is bright orange. Unlike others in the reticulatous group, Persian melons do not slip from the vine when mature.
The inodorous variant lacks the aromatic or musky odor of the reticulatous group and generally does not slip from the vine. This group includes the casaba, crenshaw, Christmas, canary and honeydew melons. The casabas are oblate in shape with bright yellow, ribbed rinds. The flesh is white or cream colored. Crenshaws are similar to casabas but slightly more oblong in shape, mostly smooth and green ripening to a yellow/green color. The flesh of crenshaws is pale orange. Christmas melons are football shaped with yellow- and green-mottled rinds and green flesh. Canary melons are oblong with bright yellow rinds. The flesh is cream colored and very mild—almost bland—in flavor. Canary melons are also called Spanish melons. Honeydews usually have green rinds with green or cream-colored flesh. Some honeydews may have yellow rinds.
Among the inodorous group, the honeydew melon is the most important. Honeydew melons have a fine pubescence that is not readily visible but can be felt. When this pubescence is gone, these melons are at full maturity. In particular, crenshaw, casaba, canary and Christmas melons are referred to as winter melons. These long-season melons have excellent keeping quality and can be stored at room temperature for a month or more.
Although there are many different types of melons described here, all these melons belong to the same genus and species and therefore can interbreed readily with fertile offspring. Because of this, many intermediate types are possible and these descriptions may not apply in all cases.
Planting
Soil should be deeply turned with previous crop residues buried at least two to three weeks prior to planting. This will allow sufficient time for previous crop residues to decompose. Any preplant fertilizer should be applied before final soil preparation (see the following section). For direct seeding, the soil should be prepared so there is a smooth debris- and clod-free surface into which the seed can be planted.
Several different planting schemes can be used to establish cantaloupes and related melons. Plant spacing can range from 4 to 6 feet between rows and 2 to 3 feet in the row. Spacing will be determined by several factors, not the least of which is the available equipment and its size. This will largely determine between-row spacing. In-row spacing also will be determined in part by available equipment. In addition to equipment, factors such as plastic mulch use, trickle irrigation installation and fertigation employment will determine in-row spacing. Plastic mulch with under-plastic trickle irrigation and fertilizer injection will allow for much closer in-row spacing (2 feet) because water and fertility can be more closely monitored and better managed.
Soils and Fertilizer Management
Soil Testing
It?s impossible to make a blanket lime and fertilizer recommendation that would be adequate for all cantaloupe fields. Each field is different, and every growing season seems to be unique.
In addition to crop nutrient requirements and general soil types, fertilizer recommendations are influenced by soil pH, cation exchange capacity (the ability of a soil to hold nutrients), residual nutrients, and inherent soil fertility. Fertilizer recommendations based on soil testing have the most potential for providing cantaloupes with adequate, but not excessive, nutrient levels. Application of optimum amounts of fertilizer results in the best yield and doesn?t encourage luxury consumption or cause fertilizer burn. Please keep in mind, however, that recommendations based on soil test analyses are valid only if proper soil sampling procedures are used. To be beneficial, a soil sample must reliably represent the field or management unit from which it was taken. Improperly collected, compiled, handled or labeled soil samples may be detrimental.
Before each cropping season, soil tests should be taken to determine fertilizer needs. University of Georgia soil sampling bags may be obtained from local county Extension offices. Please contact your local county Extension office if you have questions about soil sampling procedures.
Soil pH
Soil pH influences the availability of nutrients in the soil, the activities of soil microorganisms and, subsequently, plant growth, yield and cantaloupe quality. Maintaining soil pH in the 6.0 to 6.5 range is important for cantaloupe production. A soil test indicates the soil pH and recommends the amount of lime, if any, that should be applied to raise the pH to the desired level.
Because calcium is not very mobile in the soil, lime should be broadcast and thoroughly incorporated 6 to 8 inches deep to neutralize the soil acidity in the root zone. For best results, lime should be applied and incorporated 2 to 3 months before seeding or transplanting.
The liming materials commonly available in Georgia are calcitic and dolomitic limestone. Because dolomitic limestone also contains 6 percent to 12 percent magnesium, it is usually the preferred liming material for soils needing magnesium.
Fertilization
Phosphorus and Potassium Recommendations
The following chart indicates the pounds of fertilizer nutrients recommended for varying soil fertility levels according to soil test ratings of residual phosphorus (P2O5) and potassium (K2O).
Table 1. Fertilizer Recommendations for Cantaloupe
| Ratings | Low | Medium | High | Very High |
| Recommended phosphorus | 120 | 80 | 40 | 0 |
| Recommended potassium | 120 | 90 | 60 | 30 |
Phosphorus ? represents pounds of P2O5 recommended per acre; Potassium ? represents pounds of K2O recommended per acre.
NOTE: If soil testing is done by a lab other than the University of Georgia Soils Testing Laboratory, the amounts recommended above may not apply.
Phosphorus
All the recommended phosphorus should be applied during or near seeding or transplanting. If only small amounts of fertilizer phosphorus are needed, banding it is usually best because phosphate moves very little in the soil.
If transplanting cantaloupes, apply approximately 0.5 pint of a starter solution consisting of 3 pounds of
10-34-0 (or similar material) mixed in 50 gallons of water to stimulate rapid growth of cantaloupe transplants. For seeded or transplanted cantaloupes, apply 100 to 150 pounds per acre of a pop-up fertilizer such as 10-34-0 to enhance early growth, especially in cool/cold soils early in the spring. Pop-up fertilizer should be banded 2 to 3 inches to the side of the row and 2 to 3 inches below the seeds or roots.
Potassium
One-third to one-half of the potassium should be either (1) applied in two bands, each located 2 to 3 inches to the side and 2 to 3 inches below the level of seed or plant roots, or (2) incorporated in the bed prior to transplanting. Research has shown that broadcasting over the entire field is usually less effective than banding.
An alternative to field broadcasting is the modified broadcast method in which the preplant fertilizer containing nitrogen, potassium and any recommended micronutrients are broadcast in the bed area only. For example, when cantaloupes are seeded or transplanted on 72-inch centers, a swath (30 to 48 inches wide) of fertilizer is uniformly applied centered over the bed. Incorporation by rototilling helps reduce water and wind movement of the fertilizer and also will place some fertilizer in the root zone. The remaining recommended potassium should be applied in one to three applications as needed. It can be banded in an area on both sides of the row just ahead of the developing root tips. The maximum number of applications is usually more effective on sandy soils.
Nitrogen Recommendations
Typical Coastal Plain soils require a total of 80 to 120 pounds of nitrogen (N) per acre. Extremely sandy soils may need additional N or an increased number of applications. Piedmont, Mountain and Limestone Valley soils usually require only 80 to 100 pounds of N per acre for cantaloupe production.
For each specific season, the required N rates may vary depending on rainfall, soil type, irrigation, plant population, duration of the harvest season, and method and timing of applications. For typical Coastal Plain soils, one-fourth to one-third of the recommended nitrogen should be either (1) applied in two bands, each located 2 to 3 inches to the side and 2 to 3 inches below the level of seed or plant roots, or (2) incorporated in the bed prior to transplanting. Research has shown that broadcasting over the entire field is usually less effective than banding. An acceptable alternative to field broadcasting is the "modified broadcast" method (see "Phosphorus" and "Potassium" above). Incorporation by rototilling will help reduce water and wind movement of the fertilizer and will also place some of the nitrogen in the root zone. Apply the remaining recommended N in one to three applications as needed. It can be banded in an area on both sides of the row just ahead of the developing root tips. For heavier Piedmont, Mountain and Limestone Valley soils, one to two applications are usually sufficient.
Magnesium, Sulfur, Zinc and Boron Recommendations
If the soil test indicates magnesium is low and if lime is recommended, apply dolomitic limestone. If magnesium is low and lime is not recommended, apply 25 pounds of elemental magnesium per acre. Apply a minimum of 10 pounds of sulfur per acre, 1 pound of actual boron per acre, and, if soil test indicates zinc is low, apply 5 pounds of actual zinc per acre.
Foliar Application of Fertilizer
In general, foliar application of major nutrients is a questionable practice. It is virtually impossible for cantaloupe plants to absorb enough nitrogen, phosphorus or potassium through their leaves to correct any major deficiencies. Foliar applications of secondary and micronutrients may be helpful in alleviating deficiencies. However, they should be applied only if there is a real need for them and only in quantities recommended for foliar application.
Two to three foliar applications of water-soluble boron (1 ounce by weight of actual boron per application) at weekly intervals coinciding with flowering can enhance fruit set if there is a boron deficiency. Many growers prefer a commercial formulation containing both boron and calcium. Be careful when applying manganese and boron to cantaloupes. Toxicity has been observed when manganese reaches 900 parts per million or when boron reaches 150 ppm in leaf tissue. Please follow manufacturer?s directions when applying any commercial foliar fertilizer formulation.
Tissue Analysis
Fertilizer recommendations provide general guidelines for the application of fertilizers to crops. Actual amounts needed will vary depending on soil type, the amount of leaching, and crop growth. Routine tissue analysis is an excellent tool for fine tuning fertilizer management. Tissue analysis should begin early in the crop cycle. County Extension offices have information on collecting a plant sample for analysis.
Table 2. Plant Tissue Analysis Critical Values for Cantaloupe
|
Status |
Nutrient |
|||||||||||
| N1 (%) |
P (%) |
K (%) |
Ca (%) | Mg (%) | S (%) |
Fe (PPM) | Mn (PPM) | Zn (PPM) | B (PPM) | Cu (PPM) | Mo (PPM) | |
| 12-inch vines2 | ||||||||||||
| Deficient | <4.0 | <0.40 | <5.0 | <3.0 | <0.35 | — | <40 | <20 | <20 | <20 | <5 | <0.6 |
| Adequate range | 4.0 5.0 |
0.40 0.70 |
5.0 7.0 |
3.0 5.0 |
0.35 0.45 |
0.2 0.5 |
40 100 |
20 100 |
20 60 |
20 80 |
5 10 |
0.6 1.0 |
| High | >5.0 | >0.70 | >7.0 | >5.0 | >0.45 | — | >100 | >100 | >60 | >80 | >10 | >1.0 |
|
Early fruit set 2 |
||||||||||||
| Deficient | <3.5 | <0.25 | <1.8 | <1.8 | <0.30 | — | <40 | <20 | <20 | <20 | <5 | <0.6 |
| Adequate range | 3.5 4.5 |
0.25 0.40 |
1.8 4.0 |
1.8 5.0 |
0.30 0.40 |
0.2 0.5 |
40 100 |
20 100 |
20 60 |
20 80 |
5 10 |
0.6 1.0 |
| High | >4.5 | >0.40 | >4.0 | >5.0 | >0.40 | — | >100 | >100 | >60 | >80 | >10 | 1.0 |
Adapted from Plant Tissue Analysis and Interpretation for Vegetable Crops in Florida. University of Florida publication SS-Vec-42. January 1991.
1. N= nitrogen, P = phosphorus, K = potassium, Ca = calcium, Mg = magnesium, S = sulfur, Fe = iron, MN = manganese, Zn = zinc, B = boron, Cu = copper, MO = molybdenum
2. Most recently mature leaf sampled
Irrigation
Water is a critical component in the production of cantaloupes and specialty melons in Georgia. Most melon crops are potentially deep-rooted (3 to 4 feet); however, in Georgia soils the effective rooting depth is generally much less. Actual rooting depth will vary considerably depending on soil conditions and cultural practices. The restricted rooting depth and the fact that melons are commonly grown in sandy soils with a low water-holding capacity make irrigation necessary for consistently high yields in Georgia.
Water deficits during the establishment of cantaloupes, etc. delay maturity and may cause gaps in production. Water stress in the early vegetative stage results in reduced leaf area and reduced yield. The most serious yield reductions result from water stress during flowering and fruit development.
Several types of irrigation systems may be used successfully on melons in the Southeast. Ultimately, the decision about which type to choose will be based on one or more of the following factors:
- availability of existing equipment
- field shape and size
- amount and quality of water available
- labor requirements
- fuel requirements
- cost
Sprinkler Irrigation
Currently, most cantaloupes are irrigated with some type of sprinkler irrigation. These systems include center pivot, linear move, traveling big gun, permanent set and portable aluminum pipe with sprinklers. Any of these systems are satisfactory if used correctly. However, initial cost and labor requirements differ significantly.
Any sprinkler system used on melons should be capable of delivering at least an inch of water every four days. In addition, the system should apply the water slowly enough to prevent runoff. With most soils, a rate less than 2 inches per hour safely prevents runoff.
Sprinkler systems with a high application uniformity (center pivot and linear move) can be used to apply fertilizer through the system. This increases the efficiency of fertilizer utilization by making it readily available to the plant and reducing leaching.
Sprinkler irrigation is not generally recommended for crops planted on plastic mulch; however, this has been a fairly common practice for watermelons and cantaloupes in Georgia.
If overhead irrigation is used with plastic mulch, it is advisable to use the narrowest bed that is practical so that water can more readily reach the crop roots. This practice is not recommended for deep sandy soils.
Drip Irrigation
Drip irrigation is gaining popularity for production of vegetable crops in Georgia. It can be used with or without plastic mulch. One of the major advantages of drip irrigation is its water use efficiency if properly managed. Studies in Florida have indicated that 40 percent less water was required for drip irrigated vegetables than sprinkler irrigated vegetables. Weeds also are less of a problem because only the rows are watered; the middles remain dry. Some studies have indicated that drip also enhances earlier yields and fruit size.
Drip tubing (or tape) may be installed on the ground surface or buried just below the surface. When used in conjunction with plastic mulch, the tape can be installed at the same time the plastic mulch is laid. Offsetting the tape slightly from the center of the bed is usually desirable. This prevents the tape from being damaged during the hole punching and the planting operation.
Typically, one line of drip tape is installed beside each row. A field with a 5-foot row spacing will require 8,712 feet of tape per acre.
The tape is available in various wall thicknesses ranging from 4 mils to 25 mils. Most growers use thin wall tape (less than 10 mils) and replace it every year. Heavier wall tape can be rolled up at the end of the season and reused; however, care must be taken in removing it from the field.
Drip systems can be easily adapted for the injection of fertilizer. This allows plant nutrients to be supplied to the field as needed. This method also eliminates the need early in the season for heavy fertilizer applications, which tend to leach beyond the reach of root systems or cause salt toxicity problems. Only water-soluble formulations can be injected through the drip systems. The system should be thoroughly flushed following each injection.
Water used in a drip irrigation system should be well filtered to remove any particulate matter that might plug the tape. The water should be tested for minerals that might cause plugging problems.
Scheduling Irrigation
The water used by a crop and evaporated from the soil is called evapotranspiration, or ET. ET rates for cantaloupes and specialty melons have been reported as high as 0.3 inches per day. Factors that affect ET are stage of crop growth, temperature, relative humidity, solar radiation, wind and plant spacing.
The following is a general recommendation for irrigation rates on sprinkler irrigated cantaloupes, etc.:
- From planting until plants begin to run apply 0.5 inch whenever soil in top 6 inches becomes dry (about every five or six days when weather is dry).
- From time plants begin to run until first bloom apply 0.75 inch every five days during dry weather. If wilting occurs before noon, increase frequency of irrigation
- From first bloom until harvest apply 0.75 inch every three days during dry weather. During extremely hot weather (over 95°), application amounts may need to be increased to 1 inch to avoid stress. Sandy soils may require more frequent, lighter applications than heavier soils to prevent moisture stress.
Another alternative to determine irrigation frequency is to use the pan evaporation coefficients in Figure 1. Multiply the coefficient by daily pan evaporation to estimate daily ET.
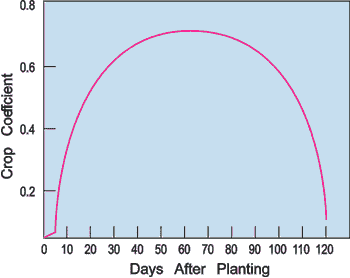 Figure 1.
Figure 1.Drip irrigation systems need to be operated more frequently than sprinkler systems. Typically, they are operated every day or every other day. Do not over water, especially when using plastic mulch, because the plastic will keep the soil from drying out.
Soil moisture monitoring can be used to fine tune irrigation applications. This ensures that soil moisture is adequate to prevent crop stress. The irrigation schedule should be adjusted whenever soil moisture measurements indicate overly wet or dry conditions.
Soil moisture may be monitored using either electric resistance blocks (such as the Watermark™ soil moisture sensor) or tensiometers. Install two sensors at each monitoring location: one about 6 to 8 inches deep and one 12 to 16 inches deep. Each field should have a minimum of two monitoring locations; more for fields larger than 20 acres or if soil types vary considerably.
The shallow sensors are near the middle of the root zone and will indicate when irrigation should be started. Up until first bloom, readings should not exceed 50 centibars. Afterward, they should not exceed 30 centibars. The optimum range for soil moisture is 5 to 30 centibars. The deep sensor is used to evaluate previous irrigations. If readings remain low (less than 5 centibars), then irrigation amounts should be decreased. If they continue to increase even after an irrigation, then irrigation amounts should be increased. Read soil moisture sensors at least three times per week during dry weather. For drip irrigated fields, sensors should be read even more frequently, typically every day during flowering and fruit development.
Disease Management
Cantaloupes and specialty melons are subject to attack by many diseases, which causes serious losses throughout the state each year. Because they are closely related, both crops share several common diseases. A clear understanding of the diseases and the strategies used to manage them is necessary for profitable cantaloupe and specialty melon production. Each disease affecting these crops will be discussed.
Fungal Diseases
Diseases caused by fungi are the most damaging to most crops, including cucurbits. Fungal inoculum (spores, hyphae, sclerotia, etc.) may be spread by wind, rain, infected plants and seed, and infested soil carried on machinery. As with most plant diseases, prevention is the key to disease management.
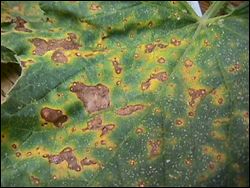 Figure 2. Alternaria leaf spot. Figure 2. Alternaria leaf spot. |
Alternaria Leaf Spot
Alternaria leaf spot is caused by Alternaria spp. and can cause serious damage under extended periods of wet weather. The occurrence of this disease in Georgia is sometimes sporadic but can be devastating if left unchecked. The disease causes tiny brown spots that enlarge on the leaves, causing a target spot with concentric rings. Older lesions will develop a dark color in the concentric pattern (Figure 2). The dark color is caused by spore production, which can cause new infection sites if no protective measures are taken. Most fungicides used in disease management will suppress Alternaria leaf spot; products containing chlorothalonil are generally the most effective. No resistant cultivars are available. |
 Figure 3. Anthracnose. Figure 3. Anthracnose. |
Anthracnose Anthracnose, caused by the fungus Glomerella cingulata var. orbiculare (Colletotrichum lagenarium, C. orbiculare), attacks all above ground parts of the cucurbit plant. The fungus causing anthracnose overwinters locally on old cucurbit vines and may appear any time during the growing season. It may reach epidemic proportions when rainfall is above average and temperatures are between 70° and 80°F. Oldest leaves exhibit the first symptom of anthracnose: round, reddish-brown spots. The centers of some spots fall out, giving the leaf a "shot-hole" appearance (Figure 3). Often the leaves at the center of the plant are killed first, leaving the crown of the plant bare. Light brown to black elongated streaks develop on stems and petioles. Round, sunken lesions may appear on the fruit. These lesions are first water-soaked and then turn a dark green to brown. The pinkish ooze often noticed in the center of the lesion is a mass of spores of the fungus. A one-year rotation and deep turning infected debris immediately after harvest are effective cultural practices for reducing inoculum levels in subsequent crops. Using disease-free seed produced from areas not known to have anthracnose is an essential disease prevention measure. Cultivars resistant to anthracnose races 1, 2 and 3 may be used to prevent disease losses. Several protectant fungicide options are available and can be found in the Georgia Pest Control Handbook. |
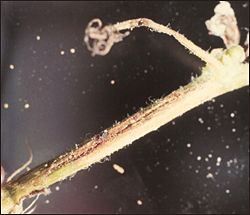 Figure 4. Gummy stem blight. Figure 4. Gummy stem blight. |
Gummy Stem Blight Gummy stem blight, caused by Didymella bryoniae, attacks primarily the leaves and stems of cucurbits and is one of the most destructive diseases of cucurbits in the state. This disease is driven by cool moist periods, especially extended periods of leaf wetness. The gummy stem blight fungus can easily be brought into a new area on or in the seed. Once the disease becomes established, it produces millions of sticky spores. These spores are spread over the field as people, animals and machines move through wet vines. It is noticeable when an individual runner or an apparently healthy plant suddenly dies. Vine cankers are most common near the crown of the plant. This disease is usually identified by finding elongated, water-soaked areas on the stems of cucurbits. These areas become light brown cracks in the vine and usually produce a gummy ooze (Figure 4). On the older leaves this disease may produce brown to black spots. It spreads from the center of the hill outward, as do anthracnose and downy mildew. Choosing high-quality, disease-free seed and transplants should be the first line of defense in preventing losses to gummy stem blight. A two-year rotation to crops other than cucurbits is another appropriate disease management tool. Protective fungicide sprays can offer the most effective disease suppression if applied in a timely manner. Please consult the Georgia Pest Control Handbook for details concerning protective fungicides. |
 Figure 5. Target spot. Figure 5. Target spot. |
Target Spot Target spot, caused by the fungus Corynespora cassiicola, can defoliate and destroy an entire crop if left unchecked. It occurs very sporadically and can be confused with downy mildew and other leaf-spotting diseases. Target spot begins on leaves as yellow leaf flecks, which later become angular with a definite outline (Figure 5). Later spots become circular with light brown centers surrounded by dark brown margins. Lesions coalesce to produce large dead areas with dead and shedding leaves. Most protectant fungicides used to control other foliar pathogens will suppress target spot. Destroying infected debris or sanitizing greenhouse areas will greatly aid in reducing the spread of disease. Resistant cultivars, if available, should be used in conjunction with the aforementioned disease management strategies. |
 Figure 6. Fusarium wilt. Figure 6. Fusarium wilt. |
Fusarium Wilt The fungus that causes Fusarium wilt of melon, Fusarium oxysporum f.sp. melonis, specifically attacks muskmelon and occurs sporadically in the Southeast. However, it is very devastating to fields it infests. Symptoms of Fusarium wilt differ from bacterial wilt in that a general yellowing of foliage usually precedes wilting (Figure 6). However, sometimes a sudden wilt occurs without any yellowing of foliage occurs. Fusarium wilt is usually more severe later in the season because of the stress of fruit load. Vascular discoloration may or may not occur. The primary means of managing Fusarium wilt is through the use of resistant cultivars. However, there are at least four races of the pathogen, and it is necessary to determine which race of the pathogen is present before choosing a cultivar. Rotation is not very effective because the chlamydospores can survive long periods in the soil and the fungus can also survive in the roots of non-crop host plants. Broad-spectrum fumigants may reduce initial inoculum levels resulting in acceptable control. Maintaining a pH of 6.5 to 7.0 and reducing nitrogen levels may also suppress wilt. |
|
Belly rot has been a common problem for cantaloupes in Georgia. The two fungi primarily responsible for belly rot are Pythium (also called cottony leak) and Rhizoctonia. Belly rot can occur on fruit at any stage of growth; however, it is most noticeable when cucurbits are mature. Symptoms may vary from small, yellow sunken areas to large rotted spots on the undersides of fruit. Pythium causes a water-soaked lesion that develops into a watery soft rot. White, cottony mycelium is also generally associated with Pythium lesions. Rhizoctonia belly rot typically appears as dry, sunken cracks on the underside of cucurbit fruit. Rotation and deep turning are cultural practices that can reduce the amount of disease inoculum near the soil surface. Practices that ensure good drainage can also reduce losses to these fungi. Systemic fungicides may aid in suppression of Pythium; fungicides have proven to be inconsistent in dealing with Rhizoctonia belly rot. |
|
 Figure 7. Southern blight. Figure 7. Southern blight. |
Southern Blight The soilborne fungus Sclerotium rolfsii causes Southern blight of cantaloupes and melons. This disease is particularly damaging to fruit lying on moist soil during periods of warm weather. Coarse white mycelium and reddish tan sclerotia are characteristic signs of the disease (Figure 7). Infected tissue is usually water soaked and disintegrates rapidly. Extensively damaged fruit will collapse upon handling. Cultural disease management practices such as deep turning and long rotations are the most feasible and effective tactics. Other methods include avoiding damaging fruit and depositing soil over runners. Fungicidal control is rarely practical or effective. |
|
Scab, caused by the fungus Cladosporium cucumerinum, inflicts severe losses on melons in all areas where moisture is high and temperature is relatively low (70° to 75°F). Most damage occurs in North Georgia, where weather conditions are more favorable for disease development. However, damage to fall melons in South Georgia can occur. The fungus causing scab lives over winter in old cucurbit vines and on infected seed. Early spring infection may come from either of these sources. Spots are produced soon after the fungus begins to sporulate; spores are spread by insects, clothing or tools, or are blown long distances in moist air. Although scab can attack any above ground portion of the plant, injury to the fruit is most noticeable. Fruit can be infected at all stages of growth. Spots first appear as gray, sunken areas about 1/8 inch in diameter from which a sticky substance may ooze. The spots grow darker with age and gradually sink into the fruit until a cavity is formed. Several areas may run together, forming lesions 0.5 inch in diameter. The presence of pale-green water-soaked areas is the first sign of disease on the foliage. These spots gradually turn gray to white and become angular shaped. The dead leaf tissue usually splits open and falls out, leaving a ragged hole in the leaf. Under favorable weather conditions, scab can deform young leaves by shortening the internodes. Losses to this disease can be effectively and economically reduced through a complete disease control program. The critical steps are as follows:
|
|
 Figure 8. Crown rot. Figure 8. Crown rot. |
Crown Rot
Crown rot is caused by the fungus Phytophthora capsici and may cause serious damage once established. The fungus infects all above ground plant parts of cucurbits. This disease became established in 1994 following hurricane Alberto. It is thought that spores of the fungus were deposited over the entire state by the hurricane. The major damage occurred in squash and pepper the first year but, because of a relatively wide host range, the disease is now causing problems in watermelons, cantaloupes, pumpkins, cucumbers and eggplant. Entire plantings of the above mentioned crops were affected in 1997. Symptoms on cucurbits appear as constricted, water-soaked lesions near the base of stems that are in close proximity to the soil. Infected fruit may have circular, sunken, water-soaked lesions, which may contain a pasty or powdery sporulation of the fungus (Figure 8). Rotation with a non-susceptible crop has been highly effective in disease prevention. It is not advisable to plant a susceptible crop in an infested field for two years. Measures that ensure good field drainage such as using crowned beds, subsoiling and avoiding over-irrigation will lessen the severity of disease. Preventive applications of some fungicides have shown some promise. |
Mildews
Mildews are a common occurrence in commercial cucurbit plantings and cause growers to spray on a regular basis. This practice alone will result in increased yields and a higher percentage of marketable fruit. Two distinct mildew diseases exist; each favored by a different weather pattern, and each requiring different materials for control. Unfortunately, the symptoms and occurrence of these diseases overlap considerably.
Bacterial Diseases
 Figure 12. Evidence of nematodes. Figure 12. Evidence of nematodes. |
NematodesNematodes are small, slender, microscopic roundworms that live in the soil. The root-knot nematode is the most common type attacking cucurbits. If not controlled, this pest can severely damage cucurbit crops, especially on light, sandy-textured soils. Root-knot nematodes enter young cucurbit feeder roots during their common feeding process, causing the roots to swell. The most common below ground symptom is the formation of galls or knots on the roots (Figure 12). Nematode injury interferes with the uptake of water and nutrients, thus causing the plants to appear as if they lack moisture or fertilizer. Stunted, yellow, irregular growth of plants in the field and rapid decline are also above ground symptoms of nematode injury. Rotating cucurbits with a grass crop such as rye or corn is somewhat beneficial in managing root-knot nematodes, but this practice is no substitute for soil fumigation. In the light soils of South Georgia, where root-knot nematodes are widespread, the use of soil fumigants is essential in most fields if maximum yields are to be expected. Fumigant nematicides are most effective against nematodes and must be applied three weeks prior to planting. Materials, rates, and methods of application can be found in the Georgia Pest Management Handbook. |
Viral Diseases
Mosaic
One or more of five major viruses causes this disease. The most prevalent are cucumber mosaic virus (CMV), papaya ringspot virus (PRSV, formerly called WMV-1), watermelon mosaic virus (WMV, formerly called WMV-2), and zucchini yellow mosaic virus (ZYMV). The most common virus in cucurbits is CMV. One or a combination of these viruses may affect cantaloupes and melons. The primary vectors associated with these virus diseases are aphids. Aphids must acquire the virus from a host reservoir and are capable of transmitting it for 10 to 15 minutes in most cases.
Symptoms of virus disease are mottling, strapping and vein distortion. One virus may cause mild symptoms; additional viruses in the same plant cause much more dramatic symptoms. In some cases, the symptoms may appear to be phytotoxic chemical damage. Fruit from infected plants may be discolored or have raised bumps or mottles. Resistant varieties are available to some, but not all, of these viruses. Stylet oil sprayed on a two- to three-day schedule has been shown to delay the spread of virus, particularly in the fall. Viral diseases are much worse in late summer and fall because the aphid populations are much higher and virus-carrying host plants are more available, which result in more viruliferous aphids. Yield losses are directly related to time of infection. The later the infection occurs, the less damage will be observed.
Insect Management
Cantaloupe and other melons are subject to attack by a variety of insect pests. These attacks do not always result in economic injury, so certain insect management practices can be used to ensure cost-effective control decisions. Indiscriminate use of insecticides often creates more favorable conditions for the development of harder-to-control insect pests, thus increasing the cost of production.
Insects cause injury to the leaves, stems, roots and melons. The developmental stage of the plant at the time of attack often governs what plant part different insect pests may injure. However, some insects feed specifically on one plant structure while others may feed on several structures.
Certain cultural practices may have dramatic effects on the potential for economic injury by certain insects. Planting during optimum growing conditions ensures rapid seedling emergence and subsequent growth. This reduces the amount of time that plants are susceptible to injury from seedling insect pests.
Most insect problems can be treated as needed if detected early, but no one insecticide will adequately control all the insects that may attack melons. Scouting for insects is the most efficient way to determine what problems may exist and what action should be taken. Preventive treatments may be necessary for certain insect pests. Preventive treatments are used against insects that are certain to cause economic injury if they are present. Preventive treatment decisions are influenced by field history, harvest dates and insect pressure in nearby production areas.
Miscellaneous Insect Pests
Some insects are occasional pests when other hosts are not available, populations are very high or environmental conditions are just right for rapid development. Flea beetles, spider mites, leaf miners, stink bugs, leafhoppers, squash bugs, whiteflies and grasshoppers are just a few. These problems can be addressed on a case-by-case basis. Contact the local county Extension agent if there are questions on the treatment of these insects.
Honeybees
Honeybees are necessary to ensure adequate pollination; most insecticides are toxic to honey bees, so follow certain practices to prevent bee kills. Honeybees may be active from dawn to dusk. Insecticide applications should be made late in the day—after sunset if possible—after bee activity has ceased. If it is necessary to spray large acreages during the day, remove hives from the field on the preceding day. If these precautions are followed, bee kills will be kept to a minimum. Once dried on the leaf surface, the toxic effects of most insecticides are dramatically reduced.
Weed Management
Weed control is one of the most serious concerns for commercial cantaloupe and specialty melon growers. Weeds compete with the melon crop for light, nutrients, water and physical space. In addition, weeds can harbor insects, diseases and nematodes harmful to the crop. This can be especially true of many wild type melons, including gherkin, small melon, citron melon and others. Furthermore, many weed control measures will be ineffective against these weeds because of their similarity to cantaloupes and other commercial melons.
Factors Affecting Weed Control
Site selection is the first, and often the most important, step in weed management for these crops. Avoid areas with heavy infestations of perennial weeds such as Bermuda grass, Johnson grass, and purple and yellow nutsedge. Other problem weeds can include sicklepod, Florida beggarweed, morning glories, cocklebur, Texas panicum and the wild type melons mentioned previously. If these areas cannot be avoided, techniques such as stale seedbed should be employed to reduce the levels of infestations prior to planting. In the stale seedbed technique, the land is prepared as normal before planting, weeds are allowed to germinate, and then a nonselective herbicide is used to eliminate this "first flush." The crop is then planted without disturbance to the soil, because this will cause more weed seeds to germinate. In addition, many weeds will germinate before melon emergence and can be eliminated using a nonselective herbicide.
Weed identification is essential because many weed control techniques are only effective on certain weeds. For example, Poast® herbicide is very effective on most grass weeds but does not control nutsedge or nutgrass, a similar-looking species. Crop rotation is also important to maintain a minimum infestation of troublesome weeds. During the process of rotation, land treated with herbicides to which melons may be sensitive should be avoided. Many of the herbicides used for weed control in agronomic crops (e.g., peanuts, soybeans, corn, cotton, grain sorghum) have residual activity in the soil and may damage melons. Keeping a record of the herbicides used on fields to be planted is imperative, and the herbicide labels must be checked for crop rotation guidelines.
An essential component to weed management for cantaloupes and other specialty melons is the growing of a good crop. Proper fertility, water, row spacing, pest control and other cultural factors that promote a rapid-growing, high-quality crop will allow the crop to outcompete many weeds. Therefore, early season weed control is essential to maintain good growing conditions. Research has shown that weed pressure in the first five weeks of melon growth will cause at least a 20 percent loss in yield. On the other hand, melons are extremely competitive, as anyone who has tried to control wild melons in other crops can attest. In fact, research has shown that melons require a weed-free period of five weeks. After that, any amount of weed infestation does not affect yield.
Methods of Weed Control
Several weed control methods are employed in melon production, but factors such as cost, size of crop, size and type of weed infestation, and available control measures will need to be considered in establishing a weed control strategy.
Hand weeding or hoeing is often the safest and least damaging to the crop; however, only growers with small acreage and abundant labor can depend on this approach. Hand weeding or hoeing is used in many cases at a last ditch effort; however, the weeds have already damaged the crop.
Mechanical control is very effective during early growth; once plants begin to produce runners, mechanical cultivation is not practical. Tractor wheels and cultivators easily damage crops. Mechanical cultivation usually requires supplementary hand weeding for removing weeds in the rows.
Chemical control through the use of herbicides is currently limited to materials recommended by the University of Georgia Cooperative Extension Service. Chemical weed control recommendations for cantaloupes and other specialty melons may be found in the current Georgia Pest Management Handbook. A pre-emergence herbicide is recommended to provide early season weed control of many grasses and broadleaf weeds. Very few compounds exist for post-emergence weed control in cantaloupes; therefore, weed control from a pre-emergence treatment is very beneficial.
Weed control using the stale seedbed technique involves chemical weed control of emerged weeds before crop emergence. A nonselective, contact material is used primarily. The stale seedbed method often is coupled with a preplant incorporated herbicide treatment. If the crop is transplanted, this method may be used to kill emerged weeds before transplanting. On direct-seeded plantings, apply the herbicide to those weeds that have emerged after planting but before the crop has emerged.
Plastic mulch with trickle irrigation is expanding rapidly for melon production and will provide very good weed control. However, many growers will use a narrow plastic band and overhead irrigation. Any holes in the plastic that will allow light penetration will be an area for weed germination, so a small hole for transplanting or seedling is advantageous. Black plastic is the most effective mulch because it prevents light penetration for weed seed germination and/or growth. However, plastic mulches, even black plastic, are not effective for nutsedge control. It is important to remember that only the area covered with plastic will provide weed control. Areas between mulched beds should be treated only with a pre-emergence or post-emergence herbicide registered for these crops, because the root systems of these plants have the capability of extending into the treated zone.
Pesticide Application
Two types of sprayers, boom and air-assisted, are used for applying insecticides, fungicides, herbicides and foliar fertilizers. The air-assisted sprayer (Figure 21) uses a conventional hydraulic nozzle plus air to force the spray into the plant foliage. Boom sprayers (Figure 22) get their name from the arrangement of the conduit that carries the spray liquid to the nozzles. Booms or long arms on the sprayer extend across a given width to cover a swath as the sprayer passes over the field.
 Figure 21.
Figure 21. Figure 22.
Figure 22.
Pumps
Three factors to consider in selecting the proper pump for a sprayer are:
- Capacity. The pump should be of proper capacity or size to supply the boom output and to provide for agitation (5 to 7 gallons per minute per 100-gallon tank capacity). Boom output will vary depending upon the number and size of nozzles. Also, 20 percent to 30 percent should be allowed for pump wear when determining pump capacity. Pump capacities are given in gallons per minute.
- Pressure. The pump must produce the desired operating pressure for the spraying job to be done. Pressures are indicated as pounds per square inch.
- Resistance to corrosion and wear. The pump must be able to withstand the chemical spray materials without excessive corrosion or wear. Use care in selecting a pump if wettable powders are to be used because these materials will cause pump wear.
Before selecting a pump, consider factors such as cost, service, operating speeds, flow rate, pressure and wear. For spraying vegetable crops, a diaphragm pump is preferable because of serviceability and pressures required.
Nozzles
Nozzle selection is one of the most important decisions to be made related to pesticide applications. The type of nozzle determines not only the amount of spray applied but also the uniformity of the applied spray, the coverage obtained on the sprayed surfaces and the amount of drift that can occur. Each nozzle type has specific characteristics and capabilities and is designed for use under certain application conditions. The types that are commonly used for ground application of agricultural chemicals for melons are the fan and cone nozzles.
Herbicides
The type of nozzle used for applying herbicides is one that develops a large droplet and has no drift. The nozzles used for broadcast applications include the extended range flat fan, drift reduction flat fan, turbo flat fan, flooding fan, turbo flooding fan, turbo drop flat fan and wide angle cone nozzles. Operating pressures should be 20 to 30 psi for all except drift reduction and turbo drop flat fans, flooding fans and wide angle cones. Spray pressure more than 40 psi will create significant drift with flat fan nozzles. Drift reduction and turbo drop nozzles should be operated at 40 psi. Flooding fan and wide angle cone nozzles should be operated at 15 to 18 psi. These nozzles will achieve uniform application of the chemical if they are uniformly spaced along the boom. Flat fan nozzles should be overlapped 50 to 60 percent.
Insecticides and Fungicides
Hollow cone nozzles are used primarily for plant foliage penetration for effective insect and disease control and when drift is not a major concern. At pressures of 60 to 200 psi, these nozzles produce small droplets that penetrate plant canopies and cover the underside of the leaves more effectively than any other nozzle type. The hollow cone nozzles produce a cone-shaped pattern with the spray concentrated in a ring around the outer edge of the pattern. Even fan and hollow cone nozzles can be used for banding insecticide or fungicides over the row.
Nozzle Material
Various types of nozzle bodies and caps, including color-coded versions, and multiple nozzle bodies are available. Nozzle tips are interchangeable and are available in a wide variety of materials, including hardened stainless steel, stainless steel, brass, ceramic and various types of plastic. Hardened stainless steel and ceramic are the most wear-resistant materials. Stainless steel tips, with corrosive or abrasive materials, have excellent wear resistance. Plastic tips are resistant to corrosion and abrasion and are proving to be very economical for applying pesticides. Brass tips have been common but wear rapidly when used to apply abrasive materials such as wettable powders. Brass tips are economical for limited use, but other types should be considered for more extensive use.
Water Rates (GPA)
The grower who plans to use spray materials at the low water rates should follow all recommendations carefully. Use product label recommendations on water rates to achieve optimal performance. Plant size and condition influence the water rate applied per acre. Examination of the crop behind the sprayer before the spray dries will give a good indication of coverage.
Agitation
Most materials applied by a sprayer are in a mixture or suspension. Uniform application requires a homogeneous solution provided by proper agitation (mixing). The agitation may be produced by jet agitators, volume boosters (sometimes referred to as hydraulic agitators) and mechanical agitators. These can be purchased separately and installed on sprayers. Continuous agitation is needed when applying pesticides that tend to settle out, even when moving from field to field or when stopping for a few minutes.
Calibration
The procedure below is based on spraying 1/128 of an acre per nozzle or row spacing and collecting the spray that would be released during the time it takes to spray the area. Because there are 128 ounces of liquid in 1 gallon, this convenient relationship results in ounces of liquid collected being directly equal to the application rate in gallons per acre.
Calibrate with clean water when applying toxic pesticides mixed with large volumes of water. Check uniformity of nozzle output across the boom. Collect from each for a known time period. Each nozzle should be within 10 percent of the average output. Replace with new nozzles if necessary. When applying materials that are appreciably different from water in weight or flow characteristics, such as fertilizer solutions, etc., calibrate with the material to be applied. Exercise extreme care and use protective equipment when active ingredient is involved.
- From Table 3, determine the distance to drive in the field (two or more runs suggested). For broadcast spraying, measure the distance between nozzles. For band spraying, use the band width. For over-the-row or directed spraying, use the row spacing.
- Measure the time (seconds) necessary to drive the required distance with all equipment attached and operating. Maintain this throttle setting!
- With the sprayer sitting still and operating at the same throttle setting or engine RPM as used in Step 2, adjust the pressure to the desired setting. The machine must be operated at the same pressure that was used for calibration.
- For broadcast application, collect spray from one nozzle or outlet for the number of seconds required to travel the calibration distance.
- For band application, collect spray from all nozzles or outlets used on one band width for the number of seconds required to travel the calibration distance.
- For row application, collect spray from all outlets (nozzles, etc.) used for one row for the number of seconds required to travel the calibration distance.
- Measure the amount of liquid collected in fluid ounces. The number of ounces collected is the gallons per acre rate on the coverage basis indicated. For example, if you collect 18 ounces, the sprayer will apply 18 gallons per acre. Adjust applicator speed, pressure, nozzle size, etc., to obtain recommended rate. If speed is adjusted, start at Step 2 and recalibrate. If pressure or nozzles are changed, start at Step 3 and recalibrate.
Table 3. Distance to Measure to Spray 1/128 Acre
One ounce discharged equals one gallon per acre.
| Nozzle Spacing (inches) | Distance (feet) |
| 6 | 681 |
| 8 | 510 |
| 10 | 408 |
| 12 | 340 |
| 14 | 292 |
| 16 | 255 |
| 18 | 227 |
| 20 | 204 |
| 22 | 186 |
| 24 | 170 |
| 30 | 136 |
| 36 | 113 |
| 38 | 107 |
| 40 | 102 |
To determine a calibration distance for an unlisted spacing, divide the spacing expressed in feet into 340.
Example: Calibration distance for a 13-inch band = 340 / 13/12 = 313 feet
Good Agricultural Practices in the Harvest, Handling and Packing of Cantaloupes
Introduction
An abundance of research during the last decade had demonstrated that fresh fruits and vegetables play an important dietary and medical role in promoting good health. Threatening to overshadow this significant benefit is the fact that foodborne disease traced to fresh produce has increased in recent years. Specifically, in 1990 and 1991, two large outbreaks of salmonellosis (disease caused by Salmonella serotypes, which are human pathogens) were traced to imported cantaloupe. Cantaloupes have a relatively high pH: 6.0 to 6.5. This makes them ideal foods for low numbers of bacterial pathogens that might be present to multiply to infectious levels. In a joint publication, FDA and USDA have issued a guidance document called Guide to Minimize Microbial Food Safety Hazards for Fresh Fruits and Vegetables. This first ever safety publication recommends good agricultural practices common to growing, harvesting, packing and transporting fresh produce to consumers. Cantaloupe growers and shippers are urged to take a proactive role to ensuring the safety of their crops by adopting these practices.
Quality and Safety
Cantaloupe quality and safety often are perceived by consumers to mean the same thing. Good quality cantaloupe may be visually appealing and smell delicious, yet may contain pathogens or toxins that can cause illness in the consumer. Safe product, in contrast, may be discolored, overmature and unappealing, yet present no hazard to the consumer. Unfortunately, the safety of fresh cantaloupe cannot be determined by its outward appearance or condition.
Field Sanitation Program
Raw Product Safety: Ensuring cantaloupe safety begins with preventing hazards in the field. The best guarantee of a safe raw product is a proactive food safety program that has been designed and implemented to identify and prevent hazards during production and postharvest handling of these vegetables. Growers and shippers should familiarize themselves with safe production practices so they might be viewed as qualified suppliers among potential buyers. Some issues of concern during production are summarized in Table 4.
Land Use History
Grazing animals on or near cropland can introduce pathogenic (to humans) bacteria to the soil. Growers should ensure that land has not been used for animal husbandry and that it is not in proximity to animal feedlots or water runoff from grazing lands. Past improper use of pesticides can result in hazardous residues on raw product. Buyers might insist on letters of guarantee from grower/shippers that the land is suitable and safe for the crops being produced. Before planting, soil residue levels of pesticides and heavy metals should be determined.
Fertilizer Use
Incompletely composted organic fertilizers may contain pathogenic (to humans) bacteria derived from animal or human feces. If organic fertilizers are used, they must be certified that they have been completely composted so no pathogens are present. Composted sewage sludge should not be used as it may contain pathogens as well as heavy metal contamination.
Irrigation
Natural surface water (e.g., canal, lake, pond) provides enough organic matter to support the growth of bacterial pathogens. Surface water may be used but should be tested for the presence of the bacterium Escherichia coli (E. coli), which is an indicator of fecal contamination. Groundwater is less likely to harbor human pathogens but should be analyzed for heavy metal and pesticide contamination.
Overhead irrigation is more likely to spread contamination to above ground plant parts than is root-zone irrigation. Growers must be able to document answers to the following questions:
- Are irrigation practices safe?
- What is the water source?
- How is water stored?
- Are animals being raised nearby?
- What tests are performed to ensure the purity and safety of the water?
Table 4. Potential Hazards During Cantaloupe Production
| Location | Source of Problem | Solution | Documentation |
|
Land use |
Fecal contamination (source of pathogens) from animals | Prevent grazing animals or feedlots on/near production land. | Grower certification of no recent animal husbandry on land used |
| Toxic pesticide residues in soil | Review pesticide history for plant back restrictions. | Pesticide selection/
application records |
|
|
Fertilizers |
Pathogenic bacteria from organic fertilizers | Use inorganic fertilizer. | Certified test results |
| Heavy metal toxicity from sewage sludge | Use certified organic fertilizers or tested and approved sludge. | Certified test results | |
|
Irrigation water |
Pathogenic bacteria from surface water | Test/monitor water supply. | Water test results |
| Heavy metal/pesticide residues in ground water | Test/monitor water supply. | Water test results | |
| Pesticide use | Illegal/hazardous residues on product | Employ only professional, licensed applicators and monitor pesticide use. | Applicator records review; residues test |
| Hand harvesting | Fecal contamination of product | Monitor field worker personal hygiene; provide field washing/sanitizing facilities. | Training programs on worker hygiene |
| Field wagons/ trucks/harvest aids | Soil and human pathogens | Clean/sanitize frequently. | Field sanitation records |
Postharvest Handling
Harvest Quality
Field maturity of cantaloupe is based on three external quality factors: the degree of slip, condition of net and background color. Cantaloupe should be harvested at the firm-ripe stage—or three-fourths to full slip—to be commercially acceptable.
Full slip is the condition in which an abcession layer has formed that will allow the whole stem of the vine to separate or slip off cleanly from the melon with a slight tug. At three-fourths slip, a fourth of the stem usually adheres and breaks rather than slipping free.
Cantaloupes will ripen after harvest but do not increase in sugar content. Cantaloupes should always be at one-half to three-quarters full slip when harvested because almost half of the final sugar concentration is accumulated a few days before this condition. A raised, well-rounded netting should be prominent and cover the entire melon, with the exception of the vein tracts.
Although cultivars vary somewhat in their color at commercial maturity, a deep uniform green indicates physiological maturity and a light yellowish tan indicates full ripeness. Avoid harvesting melons with a brown under color or browning of the vein tracts, which are signs of aging and internal softness.
Melons displaying less than three-fourths slip should not be harvested. They have not developed enough sugar to be attractive to consumers. Additionally, they are more susceptible to bruising and scuffing and subsequent decay.
Harvesting and Handling
At the peak of the season, cantaloupe must be harvested daily to reduce the number of overripe fruit. Cantaloupes are hand harvested by field crews, often with the help of several types of harvesting aids.
One type is a transverse conveyor system that allows workers to place harvested melons on a moving belt that feeds directly into a loading vehicle.
Another type is a tractor-mounted three-point hitch to which is attached an open metal trough called a sink. Workers follow along behind it, placing harvested melons into the trough as it is pulled through the field between rows. Once loaded, pickers typically hurl melons on the ground to waiting loaders who straddle the side of a flatbed wagon or truck. After catching, melons are dropped onto a hard wooden or metal surface.
Field research has demonstrated that cantaloupe bruising and cracking can occur if the drop height is greater than 8 inches onto these hard surfaces. Growers should pad these areas with insulated carpet or hard foam materials to reduce impact damage.
Cantaloupes are subject to sun scald when left unprotected in the sun after harvest. Early morning harvesting and shading with a tarpaulin will help prevent this injury. Some growers have resorted to harvesting at night using floodlights mounted on equipment.
Pressure bruising, which causes discolored internal flesh, results when cantaloupe are stacked in bulk over six to eight layers deep or when they are transported over rough roads.
Packinghouse Operations
Loaded field vehicles should be parked in the shade while waiting to be dumped at the packinghouse. This will prevent melons from warming and sunburning.
Cantaloupe can be unloaded by hand, dry-dumped onto sloping wooden ramps or wet-dumped into tanks of water to reduce physical injury. Dump tank water needs to be chlorinated at a rate of 150 PPM free chlorine. Considerable mechanical damage occurs in dry-dumping operations, including bruising, scratching, abrading and splitting. The drop height between the loaded vehicle and ramp surface should be minimized and the surface padded with insulated carpet or hard foam. In dry-dump operations, cantaloupes are sorted (to remove culls and melons showing mechanical damage), graded (based on USDA quality grade standards or buyer?s specifications), sized (using diverging metal rollers) and packed into bulk bins or shipping boxes. Alternately, some cantaloupes are elevated onto a metal roller conveyor from a dry-dump pit, then past spray washers allowing wet brushes to remove field debris. This water should be kept clean by chlorinating at 75-100 PPM free chlorine. Next, melons are graded, pass through a hot water sanitizer, diverging roll sizers and finally to pack-out bins. Water temperature in the sanitizing tank is maintained at 140°F. This system allows the melon rind to be surface sanitized to reduce the number of plant and human pathogens that may be present.
Quality Defects and Pack Specifications
External indices of quality for cantaloupe include a well-shaped, nearly spherical melon, uniform in appearance, with an absence of physical injuries (e.g., scars, sunburn, bruising, abrasions). Melons should be firm, feel heavy for their size, and exhibit a smooth stem end with no adhering peduncle (stem attachment), which suggests premature harvest.
The concentration of soluble solids (sugars) is the most reliable index to internal quality (sweetness and flavor). The current acceptable standard for determining soluble solids is a calibrated refractometer that measures °Brix (equal to percent sugar).
U.S. grade standards for cantaloupe are Fancy, No. 1, Commercial and No. 2. Distinction among grades is based predominately on external appearance and measured soluble solids. Federal standards specify a minimum of 11 percent soluble solids for U.S. Fancy, 10 percent for U.S. No. 1 and Commercial, and 9 percent for U.S. No. 2. According to USDA inspection procedures, the sampling plan for determining soluble solids content in a car lot of cantaloupe is to select seven melons at random for testing. Tolerances by count for U.S. No. 1 should not exceed 12 percent total defects, including 8 percent for permanent defects and 6 percent for serious damage, which includes not more than 4 percent serious damage by permanent defects and 2 percent for decay. For a more complete description of quality standards and grading, contact the USDA Agricultural Marketing Service or check the Internet at www.ams.usda.gov/.
Most buyers will accept only cantaloupe equivalent of U.S. No. 1 or higher. They may have their own sampling plan for determining soluble solids content in the melons. Cantaloupe are hand packed into shipping containers. Sizing is based on count per 40- to 42-pound fiberboard container or wirebound crate. Typically, nine, 12 and 15 melons per container are packed out; occasionally, 18 or 23 melons per carton.
Cooling and Storage
Cantaloupes must be precooled shortly after harvest to maintain sugar levels and flavor intensities. Traditionally, cantaloupe have been cooled using cold air, cold water or ice. The most practical means to use in Georgia is forced air or tunnel cooling. To prevent quality loss due to dehydration, the refrigeration system of the forced air cooling unit should be designed with a large evaporator coil surface to ensure high relative humidity (90 percent to 95 percent) in the moving cold air.
Under most circumstances, cantaloupe cannot be properly cooled by merely placing palletized shipping containers in a room cooler with circulating fans only. Cantaloupe boxes cannot be cooled by placing them directly on a refrigerated truck. Optimum storage requirements for cantaloupe are 36° to 41°F and 95 percent relative humidity. Under these conditions, shelf life is approximately 14 days. Storage at a temperature of 35.6°F or less will result in chilling injury seen after several days. Sensitivity to chilling injury decreases as melon maturity and ripeness increase. Full slip melons can tolerate 35°F for up to 14 days before signs of chilling injury appear. Symptoms of chilling injury include pitting or sunken areas, failure to ripen, off flavors and increased surface decay.
Mixed Load and Storage Compatibility
Cantaloupe produce high levels of ethylene (10 to 100 PPM per hour) per kilogram. Although there are several vegetables (e.g., snap beans, squash, peppers) that have approximately the same storage temperature requirements as cantaloupe, these are ethylene sensitive and therefore cannot be stored with melons. Because cantaloupe produce moderate amounts of exogenous ethylene, even at low temperature, over ripening may be a problem during distribution and short-term storage.
Postharvest Decay
Disease can be an important source of postharvest loss in cantaloupes. Postharvest decay organisms normally do not enter the product through healthy exterior tissue. These organisms require mechanical damage (e.g., abrasion, cracking) or weakening of tissue (bruising) before they can enter. Pathogens in other infected tissue or in contaminated water may enter the fruit. Field research has demonstrated that treatment of the rind using hot water immersion (131°F for 0.5 to 1 minute) can be effective in preventing surface mold and other pathogen presence. Use of this technique, however, has not gained extensively in commercial operations.
Sanitary Guidelines for Packinghouse Operations
Receiving Incoming Product
Harvest crews should remove as much dirt and mud from the product as is possible before the produce leaves the field. An area should be set aside in the receiving yard so pallets can be cleaned before dumping in bins or cooling.
Water Sanitation
Water used in cleaning and cooling should be chlorinated at a concentration of 75 to 150 PPM of free chlorine. Chlorination can be accomplished using a gas injection system, adding bleach or using calcium hypochlorite tablets. Chlorination levels in the water should be monitored frequently during operation through the use of chlorine litmus paper or, more accurately, with a chlorine test kit. Water pH should be maintained between 6.5 and 7.5 to avoid having to use excess chlorine in order to maintain recommended free chlorine levels. Excessive use of chlorine can result in excess chlorine gas, which can irritate a worker?s skin and respiratory tract, is corrosive to equipment, and increases sanitation cost.
Employee Hygiene
Good employee hygiene is very important. Employee training, health screening and constant monitoring of packinghouse sanitation practices (hand washing, personal hygiene) are important in reducing contamination by employees.
Packinghouse Equipment
Packinghouse equipment should always be maintained in clean condition. The remnants of product left on belts, tables, lines and conveyors could provide a source for microbial growth; therefore, cleaning by scrubbing to remove particles should be part of the cleaning procedure.
If it is deemed appropriate, sanitizing with a chlorine solution could be accomplished, especially on belt conveyors and equipment, by spot spraying with hand sprayers. Knives, blades, boots, gloves, smocks and aprons should be cleaned or replaced as needed.
Pest Control
A pest control program should be in place to reduce, as much as possible, the risk of contamination by rodents or other animals. In an open or exposed packinghouse operation, the best control is constant vigilance and elimination of any discovered animals and their potential nesting locations. Product and product remnants will attract pests; therefore, the daily cleaning of the packinghouse to eliminate the attractive food source should help in reducing pest activity.
Facility Sanitation
Packinghouse facilities have the potential for developing microbial growth on walls, tunnels, ceilings, floors, doors and drains. Scheduled wash down and/or sanitizing of the facility will reduce the potential for microbial growth. The cooling system should be monitored and cleaned as necessary depending on the type of system.
Temperature Control
Maintenance of proper holding room temperature could affect product quality and could be a factor in reducing microbial growth. Temperature should be monitored to ensure maintenance at established product temperature parameters.
Production Costs
Enterprise budgets may be used to estimate cantaloupe production costs and break-even prices. The cost estimates included in the budgets should be for those inputs deemed necessary to achieve the specified yields over a period of years.
Production practices, size of operation, yields and prices vary among farms, regions and times of the year. For these reasons, each grower should adapt budget estimates to reflect his or her particular situation. The budgets below are estimated for two methods of production. Conventional (bareground) production is still the dominant method, but plasticulture is becoming more important. Detailed printed and computerized budgets are available in most county Extension offices as well as on the World Wide Web.
Type of Costs
Total costs of producing any crop include both variable and fixed costs. The variable or operating costs vary with the amount of crop produced. Common variable costs include seed, fertilizer, chemicals, fuel and labor. Fixed costs include items such as equipment ownership (depreciation, interest, insurance and taxes), management and general overhead costs. Most of these costs are incurred even if little or no production takes place and are often overlooked for planning purposes.
Variable costs are further broken down into preharvest and harvest operations in the budget. This provides the grower an opportunity to analyze the costs at different stages of the production process.
Land cost may either be a variable or a fixed cost. Even if the land is owned, a cost is involved. Land is included as a variable cost in this budget. If land is doubled-cropped, each enterprise should be charged half the annual rate. Ownership costs for tractor and equipment (depreciation, interest, taxes, insurance and shelter) are included as a fixed cost per hour of use. Overhead and management are calculated by taking 15 percent of all preharvest variable expenses. This figure compensates for management and farm costs that cannot be allocated to any one specific enterprise. Overhead items include utilities, pickup trucks, farm shop and equipment, and fees.
Cost per Unit of Production
The cost categories (tables 5, 6, and 8) are broken down in cost per unit at the bottom of the budget. The preharvest variable costs and the fixed costs decline fairly rapidly with increases in yields.
Table 5. Costs per Hundredweight, 1998
| Bareground | Plastic | |
| Preharvest cost | $2.31 | $3.51 |
| Harvest and marketing cost | $1.17 | $1.17 |
| Fixed cost | $1.00 | $1.16 |
| Total cost | $4.48 | $5.84 |
(For current cost estimates, see most recent Extension vegetable budgets.)
Budget Uses
In addition to estimating the total costs and break-even prices for producing cantaloupes, other uses can be made of the budgets.
Estimates of the cash costs (out-of-pocket expenses) provide information on how much money needs to be borrowed. The cash cost estimates are most beneficial in preparing cash flow statements.
In share leases, the landlord and tenant can use the cost estimates by item to more accurately determine an equitable share arrangement.
Risk Rated Net Returns
Because yields and prices vary so much from year to year, an attempt has been made to estimate the "riskiness" of producing cantaloupes. Five different yields and prices are used in calculating risk. The "expected" values are those prices and yields a particular grower would anticipate to exceed half the time (half the time he would anticipate not to reach these values). Averages can be used for the expected values. "Optimistic" values are those prices and yields a grower would expect to reach or exceed one year in six. The "pessimistic" values are poor prices and yields that would be expected one year in six. The "best" and "worst" values are those extreme levels that would occur once a lifetime (1 in 48).
The risk rated section (Table 7) shows that with bareground production there is a 82 percent chance of covering all costs. One-half the time, the budgeted grower would expect to net $607 or more. Half the time, he would expect to net less than $607. One year out of six he would expect to make more than $1,260 per acre; another year, to lose more than $63.
The risk rated section of the plastic budget (Table 9) shows that there is a 76 percent chance of covering all costs. One half of the time, the budgeted grower would expect to net $679 or more. Half of the time, he would expect to net less than $679. One year out of six he would expect to make more than $1,604 per acre; another year, to lose more than $295.
Readers should recognize the examples shown here are estimates. They should serve as guides for developing their own estimates.
Table 6. Cantaloupe Prices, Costs and Revenue Estimates (Bareground)
| Best | Optimistic | Median | Pessimistic | Worst | |
| Yield (cwt.) | 325 | 250 | 175 | 100 | 0 |
| Price per cwt. | 12.50 | 10.25 | 8.00 | 5.75 | 3.50 |
| Item | Unit | Quantity | Price | Dollars per acre |
|
Variable costs |
||||
|
Cwt. | 10.00 | 8.00 | 80.00 |
|
Application | 6.00 | 10.00 | 60.00 |
|
Acre | 1.00 | 15.70 | 15.70 |
|
Application | 2.00 | 6.70 | 13.40 |
|
Dollar | 384.69 | 10.5% | 20.20 |
|
Application | 3.00 | 4.26 | 12.78 |
|
Acre | 1.00 | 18.00 | 18.00 |
|
Acre | 1.00 | 0 | 0 |
|
Ton | .50 | 26.00 | 13.00 |
|
Acre | 1.00 | 19.68 | 19.68 |
|
Acre | 1.00 | 51.00 | 51.00 |
|
1,000 | 3.50 | 21.75 | 76.13 |
|
Acre | 1.00 | 25.00 | 25.00 |
|
404.89 | |||
|
Cwt. | 175 | 1.17 | 204.75 |
|
609.64 | |||
|
Fixed costs |
||||
|
Acre | 1.00 | 0 | 0 |
|
Acre | 1.00 | 113.67 | 113.67 |
|
Dollar | 404.89 | 0.15 | 60.73 |
|
174.40 | |||
| Total budgeted cost per acre | 784.04 | |||
|
Costs per cwt. |
||||
|
2.31 | |||
|
1.17 | |||
|
1.00 | |||
| Total budgeted cost per cwt. | 4.48 | |||
Table 7. Cantaloupe Risk Rated Net Return (Bareground)
Net return levels (top row), the chances of obtaining this level or more (middle row) and chances of obtaining this level or less (bottom row)
|
Optimistic |
Expected |
Pessimistic |
|||||
| Returns ($) | 1,586 | 1,260 | 933 | 607 | 272 | -63 | -398 |
| Chances | 7% | 16% | 31% | 50% | |||
| Chances | 50% | 31% | 16% | 7% | |||
- Overall chance of profit is 82%.
- Expected value of net returns is $616.
- Profit is return to risk and all "zero" items in the budget.
Table 8. Cantaloupe Prices, Costs and Revenue Estimates (Plastic)
| Best | Optimistic | Median | Pessimistic | Worst | |
| Yield (cwt.) | 410 | 310 | 210 | 110 | 0 |
| Price per cwt. | 12.50 | 10.25 | 8.00 | 5.75 | 3.50 |
| Item | Unit | Quantity | Price | Dollars per acre |
|
Variable costs |
||||
|
Acre | 1.00 | 120.00 | 120.00 |
|
Application | 6.00 | 10.00 | 60.00 |
|
Acre | 1.00 | 6.00 | 6.00 |
|
Application | 1.00 | 7.50 | 7.50 |
|
Dollar | 700.41 | 10.5% | 36.77 |
|
Application | 6.00 | 4.37 | 26.22 |
|
Acre | 1.00 | 20.00 | 20.00 |
|
Acre | 1.00 | 0 | 0 |
|
Ton | .50 | 26.00 | 13.00 |
|
Acre | 1.00 | 18.76 | 18.76 |
|
Acre | 0 | 51.00 | 0 |
|
1,000 | 3.50 | 38.00 | 133.00 |
|
Roll | 2.80 | 68.50 | 191.80 |
|
Acre | 1.00 | 28.00 | 28.00 |
|
1,000 | 3.50 | 21.75 | 76.13 |
|
737.18 | |||
|
Cwt. | 210 | 1.17 | 245.70 |
|
982.88 | |||
|
Fixed costs |
||||
|
Acre | 1.00 | 86.89 | 86.89 |
|
Acre | 1.00 | 0 | 0 |
|
Acre | 1.00 | 45.09 | 45.09 |
|
Dollar | 737.18 | 0.15 | 110.58 |
|
242.56 | |||
| Total budgeted cost per acre | 1,225.44 | |||
|
Costs per cwt. |
||||
|
3.51 | |||
|
1.17 | |||
|
1.16 | |||
| Total budgeted cost per cwt. | 5.84 | |||
Table 9. Cantaloupe Risk Rated Net Return (Plastic)
Net return levels (top row), the chances of obtaining this level or more (middle row) and chances of obtaining this level or less (bottom row)
|
Optimistic |
Expected |
Pessimistic |
|||||
| Returns($) | 2,066 | 1,604 | 1,142 | 679 | 192 | -295 | -783 |
| Chances | 7% | 16% | 31% | 50% | |||
| Chances | 50% | 31% | 16% | 7% | |||
- Overall chance of profit is 76%.
- Expected value of net returns is $762.
- Profit is return to risk and all "zero" items in the budget.
Cantaloupe Marketing
Cantaloupe consumption is rising, like that of many fresh vegetables and melons. Annual per capita consumption in the mid-1970s was about 5.5 pounds. It rose to more than 8 pounds per person in the mid-1980s and to more than 10 pounds in the late 1990s.
Annual U.S. cantaloupe production peaked at 2.17 billion pounds in 1989; then did not equal or exceed this level until 1996.
Imports and exports both are growing. Mexico is the primary source for U.S. imports; Canada is the primary export market (see Table 10).
Complete production data for cantaloupes are not available. Thus, other types of data must be used to substitute for production estimates. Acreage data are available for some states, and shipment and price data are available for some. Acreage and yield data are reported for Georgia but not shipments and prices. Table 11 shows that California, Arizona and Texas account for more than 80 percent of the reported acreage. Georgia ranks fourth. Because shipments are not reported, market arrival data (Table 12) in the major markets in the United States are used to show the relative importance of producing states during each month of the year. Georgia?s primary season is June and July, with 4 percent and 2 percent of the U.S. volume respectively.
In the past, Georgia produced mostly an eastern type cantaloupe that was not graded, cooled or packed. Its sweet taste made it a good melon for consumers, but its relatively short shelf life did not make it a good product for chain buyers. The eastern type cantaloupe is marketed mostly in the South. Of the reporting markets, Atlanta receives two-thirds of reported arrivals. Columbia and Baltimore receive about 15 percent to 20 percent of arrivals. The Northeast markets receive only about 12 percent to 15 percent of annual arrivals.
In addition to longer shelf life, chains are demanding more produce be packed and palletized. The western type melon is the one preferred by the chain stores. To meet the chains? demand, Georgia growers have been producing more of the western type melons. Growers who can consistently provide premium quality melons and full marketing services (including grading, forced-air cooling, custom packing and sales direct to retail buyers) can stabilize seasonal prices and maximize their market position throughout the entire marketing period.
Table 10. U.S. Cantaloupe: Supply, Utilization and Price, Farm Weight, 1973-97
|
Year |
Supply |
Utilization |
Season-average price |
||||||||
|
Production1 |
Imports2 | Total |
Exports2 |
Domestic | Per capita use | Current dollars3 |
Constant |
||||
|
Million pounds |
Pounds |
$/cwt |
|||||||||
| 1973 | 1,130.2 |
157.5 |
1,287.7 |
--- | 1,287.7 | 6.1 |
8.08 |
22.82 | |||
| 1974 | 972.0 |
168.2 |
1,140.2 |
--- | 1,140.2 | 5.3 |
9.99 |
25.95 | |||
| 1975 | 985.8 |
138.9 |
1,124.7 |
--- | 1,124.7 | 5.2 |
10.40 |
24.64 | |||
| 1976 | 1,014.0 |
141.0 |
1,155.0 |
--- | 1,155.0 | 5.3 |
11.00 |
24.66 | |||
| 1977 | 1,089.9 |
182.8 |
1,272.7 |
--- | 1,272.7 | 5.8 |
10.60 |
22.36 | |||
| 1978 | 1,331.8 |
195.5 |
1,527.3 |
62.0 | 1,465.3 | 6.6 |
9.64 |
18.90 | |||
| 1979 | 1,242.1 |
194.6 |
1,436.7 |
59.6 | 1,377.1 | 6.1 |
11.30 |
20.43 | |||
| 1980 | 1,224.2 |
169.9 |
1,394.1 |
62.7 | 1,331.4 | 5.8 |
13.60 |
22.52 | |||
| 1981 | 1,334.6 |
138.0 |
1,472.6 |
65.5 | 1,407.2 | 6.1 |
14.10 |
21.40 | |||
| 1982 | 1,682.4 |
182.5 |
1,864.9 |
83.7 | 1,781.2 | 7.7 |
10.15 |
14.48 | |||
| 1983 | 1,453.7 |
166.1 |
1,619.8 |
87.8 | 1,532.0 | 6.5 |
11.70 |
16.01 | |||
| 1984 | 1,651.6 |
246.7 |
1,898.3 |
86.5 | 1,811.8 | 7.7 |
12.25 |
16.14 | |||
| 1985 | 1,874.3 |
246.0 |
2,120.3 |
100.4 | 2,020.0 | 8.5 |
9.45 |
12.05 | |||
| 1986 | 2,056.2 |
319.9 |
2,376.1 |
105.8 | 2,270.3 | 9.4 |
10.50 |
13.03 | |||
| 1987 | 2,027.3 |
300.8 |
2,328.1 |
107.1 | 2,221.0 | 9.1 |
9.80 |
11.79 | |||
| 1988 | 1,691.6 |
327.0 |
2,018.6 |
93.2 | 1,925.4 | 7.9 |
15.45 |
17.94 | |||
| 1989 | 2,171.4 |
476.2 |
2,647.6 |
84.1 | 2,563.5 | 10.4 |
12.55 |
13.99 | |||
| 1990 | 1,856.7 |
530.3 |
2,387.0 |
78.8 | 2,308.1 | 9.2 |
14.65 |
15.65 | |||
| 1991 | 1,664.0 |
602.5 |
2,266.5 |
75.7 | 2,190.8 | 8.7 |
12.25 |
12.59 | |||
| 1992 | 1,811.1 |
481.9 |
2,293.0 |
115.9 | 2,177.1 | 8.5 |
13.90 |
13.90 | |||
| 1993 | 1,909.7 |
458.1 |
2,367.8 |
116.2 | 2,251.7 | 8.7 |
15.60 |
15.20 | |||
| 1994 | 1,824.6 |
523.9 |
2,348.5 |
112.7 | 2,235.8 | 8.6 |
16.50 |
15.73 | |||
| 1995 | 1,927.8 |
613.1 |
2,540.9 |
118.1 | 2,422.9 | 9.2 |
18.20 |
16.91 | |||
| 1996 | 2,211.9 |
740.8 |
2,952.7 |
126.8 | 2,825.9 | 10.6 |
18.10 |
16.50 | |||
| 1997f | 2,240.0 |
675.0 |
2,915.0 |
127.0 | 2,788.0 | 10.4 |
--- |
--- | |||
1 Source: National Agricultural Statistics Service, USDA. Production data were estimated by ERS for 1982-91.
2 Source: Bureau of the Census, U.S. Department of Commerce.
3 Source of season-average price data is NASS, USDA, except 1982-91, which was estimated by ERs using state data.
4 Constant-dollar prices were calculated using the GDP implicit price deflator, 1992=100.
Table 11. Cantaloupes for Fresh Market: Area Planted and Yield by State and United States, 1995-97
|
State |
Area Planted (acres) |
Yield per Acre (cwt) |
||||
| 1995 | 1996 | 1997 | 1995 | 1996 | 1997 | |
| California | 59,300 | 59,000 | 65,500 | 190 | 230 | 255 |
| Arizona | 16,000 | 17,700 | 20,500 | 120 | 200 | 220 |
| Texas | 12,900 | 12,600 | 12,500 | 200 | 220 | 220 |
| Georgia | 6,500 | 7,500 | 10,000 | 187 | 212 | 207 |
| Indiana | 3,500 | 3,500 | 3,800 | 160 | 175 | 130 |
| Colorado | 2,000 | 2,000 | 2,000 | 90 | 120 | 110 |
| Maryland | 1,800 | 1,900 | 1,800 | 130 | 180 | 140 |
| Pennsylvania | 1,300 | 1,100 | 1,100 | 150 | 90 | 150 |
| Michigan | 900 | 800 | 800 | 190 | 210 | 140 |
| Ohio | 490 | 470 | 450 | 215 | 125 | 165 |
| United States | 104,690 | 106,570 | 118,450 | 120 | 115 | 120 |
Source: USDA, NASS
Table 12. Monthly Cantaloupe Arrivals in 22 U.S. Cities, 1997 (millions of pounds)
| State | Jan. | Feb. | March | April | May | June | July | Aug. | Sept. | Oct. | Nov. | Dec. | 1997 | 1996 |
| California | ? | ? | ? | ? | 18.7 | 57.7 | 80.7 | 77.6 | 62.4 | 24.2 | 9.6 | 2.1 | 333 | 391.4 |
| Arizona | ? | ? | ? | ? | 3.2 | 20.1 | 13.8 | 4 | 2 | 14.1 | 11.6 | 3.8 | 72.6 | 49.5 |
| Texas | ? | ? | ? | ? | 2.4 | 5.4 | 3.2 | 0.8 | 1.2 | 1 | 0.7 | ? | 14.7 | 29.5 |
| Florida | ? | ? | ? | 1.4 | 5.2 | 2.1 | ? | ? | ? | ? | ? | ? | 8.7 | 5.8 |
| Georgia | ? | ? | ? | ? | ? | 4.5 | 2.4 | 0.4 | ? | 0.2 | ? | ? | 7.5 | 6.9 |
| South Carolina | ? | ? | ? | ? | ? | 2.1 | 3.4 | 0.7 | 0.1 | ? | ? | ? | 6.3 | 4.2 |
| Maryland | ? | ? | ? | ? | ? | ? | 2.9 | 2.1 | 0.1 | 0.1 | ? | ? | 5.2 | 2.3 |
| Indiana | ? | ? | ? | ? | ? | ? | 0.9 | 1.7 | ? | ? | ? | ? | 2.6 | 1.3 |
| Michigan | ? | ? | ? | ? | ? | ? | ? | 1.3 | 0.7 | ? | ? | ? | 2 | 0.7 |
| Subtotal | ? | ? | ? | 1.4 | 29.5 | 91.9 | 107.3 | 88.6 | 66.5 | 39.6 | 21.9 | 5.9 | 452.6 | 491.6 |
| Imports (Total) | 30.4 | 31.9 | 46.6 | 55.1 | 45.6 | 11.2 | 0.8 | ? | ? | 5 | 12.6 | 30.5 | 269.7 | 219.1 |
| Total | 30.4 | 31.9 | 46.6 | 56.5 | 75.1 | 103.1 | 108.1 | 88.6 | 66.5 | 44.6 | 34.5 | 36.4 | 722.3 | 710.7 |
| Subtotal percentage of total | 0% | 0% | 0% | 2% | 39% | 89% | 99% | 100% | 100% | 89% | 63% | 16% | 63% | 69% |
| Georgia percentage of total | ? | ? | ? | ? | ? | 4% | 2% | 0% | ? | 0% | ? | ? | 1% | 1% |
1. "Arrivals" represents that portion of total production that is shipped to the markets that the USDA reports.
? indicates no reported arrivals.
Status and Revision History
Published on Oct 01, 1999
Published on Feb 24, 2009
Published with Full Review on Jan 03, 2014
Published with Full Review on Jan 30, 2017

 Figure 9. Powdery mildew.
Figure 9. Powdery mildew. Figure 10. Downy mildew.
Figure 10. Downy mildew. Figure 11. Angular leaf spot.
Figure 11. Angular leaf spot.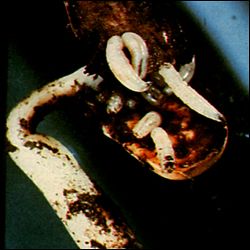 Figure 13. Root maggots.
Figure 13. Root maggots. Figure 14. Wireworm
Figure 14. Wireworm Figure 14. whitefringed beetle larvae
Figure 14. whitefringed beetle larvae

 Figure 15. Spotted cucumber beetle (top), Striped cucumber beetle and stem damage (middle), cucumber beetles and foliage damage (bottom).
Figure 15. Spotted cucumber beetle (top), Striped cucumber beetle and stem damage (middle), cucumber beetles and foliage damage (bottom). Figure 16. Colony of aphids.
Figure 16. Colony of aphids.
 Figure 17. Thrips damage, seedling (top); thrips damage, mature leaf.
Figure 17. Thrips damage, seedling (top); thrips damage, mature leaf. Figure 18. Cutworms.
Figure 18. Cutworms.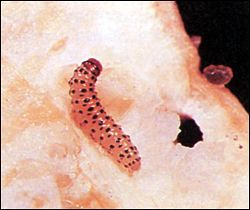
 Figure 19. Pickleworm, young larva (top); pickleworm, mature larva (bottom).
Figure 19. Pickleworm, young larva (top); pickleworm, mature larva (bottom). Figure 20. Melonworm.
Figure 20. Melonworm.