
- What Is Lime Mud?
- Does Lime Mud Have Any Harmful Components?
- How Does Lime Mud Affect Soil pH and Plant Growth?
- How Should Lime Mud Be Used?
- Are There Any Regulations Involved in the Use of Lime Mud?
- References
What Is Lime Mud?
Lime mud is a by-product produced in pulp mills as part of the process that turns wood chips into pulp for paper. The pulp mill cooks wood chips with sodium hydroxide to extract the wood fiber used to make paper from the lignin that binds the wood together. During this process, sodium hydroxide is converted to sodium carbonate. The pulp mill than adds calcium oxide, also known as quicklime, to convert the sodium carbonate back to sodium hydroxide in order to use it again. In the process, calcium carbonate is formed.
Calcium carbonate is the major component of "lime," or ground agricultural limestone. Although lime mud is largely calcium carbonate, it also contains small amounts of magnesium carbonate and other trace minerals (Table 1).
| Table 1. Typical concentrations of nutrients and minerals in lime mud from pulp and paper mills compared to agricultural lime. | ||
| Minerals | Lime Mud* | Agricultural Lime |
| Nitrogen (%) | 0 - 0.2 | 0.01 |
| P2O5 (%) | 1 - 1.2 | 0.06 |
| K2O (%) | 0.2 - 1.4** | 0.13 |
| Calcium (%) | 28 - 50** | 31 |
| CCE (%) | 91 - 100 | 80 - 100 |
| Magnesium (%) | 0.2 - 1.0** | 5 |
| Sulfur (ppm) | 0.19*** | na |
| Boron (ppm) | 7.91*** | na |
| Copper (ppm) | 3 - 66 | 10 |
| Zinc (ppm) | 4 - 93 | 113 |
| * Data from Morris, et al., 2000, except where noted. ** Data from South Carolina mills. Camberato, et al. 1997. *** Data from Alabama mills. Muse and Mitchell. 1995. na - not available |
||
Does Lime Mud Have Any Harmful Components?
Chemical analyses of lime mud have shown that it is relatively free of any contaminants. A study in Canada found organic contaminants were generally not present or found at very low concentrations. Other analyses of lime mud from mills in Georgia show the metal content is low and usually about the same or lower than that of agricultural lime (Table 2).
| Table 2. Trace metals concentrations in lime mud and agricultural lime. | ||
| Minerals | Lime Mud* | Agricultural Lime*** |
| Arsenic (ppm) | 1.71** | <1 - 3 |
| Cadmium (ppm) | bd - 0.5 | <0.1 - 1.1 |
| Lead (ppm) | bd | 1.3 - 130 |
| Mercury (ppm) | <0.05** | <0.01 - 0.02 |
| Molybdenum (ppm) | bd - 0.1 | 0.3 - 0.5 |
| Nickel (ppm) | 3.3 - 71 | 7.0 - 17 |
| Selenium (ppm) | bd - 7.6 | <1 |
| *Data from Morris, et al., 2000, except where noted. **One sample from a Georgia mill ***Data from McBride and Spiers. 2001. bd - below detection |
||
How Does Lime Mud Affect Soil pH and Plant Growth?
Soils in Georgia naturally have a low pH that can decrease crop production. Regular soil testing can determine the amount of lime needed to raise the soil pH to the desired level. For most crops in Georgia, this pH is 6.0 to 6.5.
Several studies have shown lime mud to be very effective in raising soil pH to desired levels. Lime mud applied on the surface of an Alabama pasture increased the soil pH more quickly than agricultural lime, and it was more effective that agricultural lime in raising the pH to the desired level of 6.5. Another study in the Georgia Piedmont found lime mud incorporated into the soil was as effective as agricultural lime in raising soil pH. Yields of corn, wheat, soybeans and pearl millet on test plots in Tifton and in Athens were the same or better from the lime mud plots than the agricultural limestone plots (Figure 1).
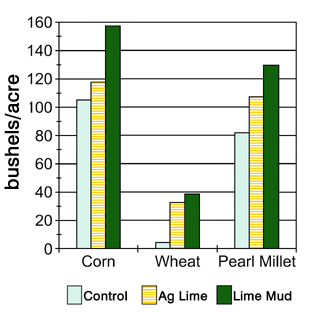 Figure 1. Crop yields (bushels/acre) using lime mud compared to agricultural lime and an unlimited control at Tifon. (Adapted from Miller and Sumner, 1999.)
Figure 1. Crop yields (bushels/acre) using lime mud compared to agricultural lime and an unlimited control at Tifon. (Adapted from Miller and Sumner, 1999.)How Should Lime Mud Be Used?
The effectiveness of lime materials is measured by the calcium carbonate equivalent or the CCE. The CCE percentage tells you how much neutralizing power each pound of that material has compared to pure calcium carbonate. A CCE near 100 percent means that a pound of the lime material has nearly the same ability to raise soil pH as a pound of pure calcium carbonate. Because the percentage of calcium carbonate in lime mud is high, the CCE of lime mud is also high, usually around 99 percent.
The particle sizes found in a lime material affect how the material can react with the soil. Materials with many fine particles raise the soil pH more quickly than those with a higher proportion of coarse particles.
Lime mud typically has a high proportion of fine particles and, consequently, reacts quickly with the soil. This means you need to treat lime mud with care to avoid high pHs that can prevent good crop growth. Agricultural limestone has coarser particles that react with the soil over a longer period of time. Lime mud reacts more like quicklime or hydrated lime and can raise the soil pH quickly. When using lime mud, take care to apply the correct amount. Over-application may raise the soil pH to levels that could create crop growth problems.
Lime mud should be applied based on soil test recommendations for lime, because the CCE of lime mud is typically near 100 percent. However, you may need to adjust for moisture content. The moisture content of lime mud is usually around 30 percent, but it can be as high as 50 percent. Lime muds with a moisture content near 50 percent may be difficult to spread, and you will need to apply more tons per acre to correct for the water weight. The formula below can be used to correct for moisture content.
Moisture Content CorrectionMoisture content can be reported as Percent Moisture or as Percent Solids. If Percent Moisture is reported, then change the Percent Moisture to a decimal by dividing by 100. Then subtract the decimal Percent Moisture from 1.00 to obtain the Percent Solids. Divide the recommended lime mud application rate by the decimal form of the Percent Solids. Example: Percent Moisture is 40%. Correct for moisture content: Lime mud application rate (tons/acre) = 2 tons/acre ÷ 0.60
If we round this number to a rate that we are able to spread, the correct application rate is 3.5 tons of lime mud per acre. |
On-site storage of lime mud should be avoided in areas where crops will be grown because it can create areas of very high pH soils. Be careful to store lime mud so runoff from the pile will not move into nearby streams or ponds.
Are There Any Regulations Involved in the Use of Lime Mud?
The Environmental Protection Division of the Georgia Department of Natural Resources regulates the land application of industrial and municipal wastes. If a by-product can be shown to have a beneficial re-use, it is not regulated as an industrial waste. Lime mud is beneficially re-used in many places as a liming material. If applied at the recommended agricultural rates, it is not considered an industrial waste by the Georgia Environmental Protection Division and can be used without having to apply for specific permits.
The Georgia Department of Agriculture requires people who distribute liming materials in the state to be licensed and to register each product. Proper labeling includes guarantees for CCE, calcium content, particle size and moisture content. It is the supplier's responsibility to see that the Department of Agriculture's requirements are followed.
In summary, lime mud is a by-product of the pulp mills that is useful as a soil liming agent. Studies have shown it is effective at raising soil pH, and crop yields are similar to those where agricultural lime has been used. Agricultural use of lime mud keeps a useful material out of landfills and may provide a lower-cost lime alternative for the farmer.
References
Camberato, J.J., E.D. Vance, and A.V. Someshwar. 1997. Composition and land application of paper manufacturing residuals. p. 185-202. In: Agricultural Uses of By-products and Wastes. ACA Symp. Ser. 668. Am. Chem. Soc.
Liard, A., R. Lessard, J. LeClerc, and L. Désilets. 1999. Product from residue: standard setting for alkaline mill residues in Quebec. Pulp & Paper Canada 100(5): 27-29.
McBride, M.B., and G. Spiers. 2001. Trace element content of selected fertilizers and dairy manures as determined by ICP-MS. Commun. Soil Sci. Plant Analysis 32 (1&2): 139-156.
Morris, L.A., W.L. Nutter, W.P. Miller, and M. Overcash. 2000. Treatment and use of pulp and paper and textile industry residues in southern U.S. forests. In: C.L. Henry, R.B. Harrison, and R.K. Bastian (eds) The Forest Alternative Principles and Practice of Residuals Reuse. Seattle, Wash: University of Washington.
Muse, J.K., and C.C. Mitchell. 1995. Paper mill boiler ash and lime by-products as soil liming materials. Agronomy Journal 87(4): 432-438.
This document was supported by the University of Georgia College of Agricultural and Environmental Sciences, Cooperative Extension Service, along with the Pollution Prevention Assistance Division.
1Extension Specialist -- Land Application, Department of Biological and Agricultural Engineering
2Professor, Department of Crop and Soil Sciences
3Professor, School of Forest Resources
Status and Revision History
Published on Mar 26, 2004
Published on Feb 23, 2009
Published on May 14, 2009
Published with Full Review on May 11, 2012
Published with Full Review on Jan 30, 2017
